1. Executive Summary of the Month
In February 2026, 111 biopharmaceutical agreements were announced worldwide. In China’s biotech sector, there were 15 out-licensing deals, 5 in-licensing deals, and 11 domestic partnerships.
In China, the top out-licensing deal involved Innovent Biologics and Eli Lilly for a preclinical asset indicated for oncology and immunology, with a total deal value of $8.85 billion and an upfront payment of $350 million. The top domestic deal was between Micot Technology and Everest Medicines for the Phase III asset MT1013, with a total deal value of $172.5 million and an upfront payment of $28 million.
Internationally, 80 cross-border deals were reported, with the largest being the agreement between Vir Biotechnology and Astellas Pharma for the Phase I/II asset AMX-500, which included an upfront payment of $240 million and a total deal value of $1.63 billion.
2026年2月,全球医药市场共签署了111项资产授权和合作协议。中国市场共达成31项交易,包括15项出海交易,5项引进交易和11项国内交易。
国内市场上,本周最大的出海交易是信达生物与Eli Lilly就肿瘤及免疫领域创新药物达成全球研发合作与许可协议,首付款3.5亿美元,总金额可达80.85亿美元。本月最大的国内交易是云顶新耀引进麦科奥特临床三期资产MT1013中国及亚太区(日本除外)的独家商业化权益,首付款2800万美元,总金额可达1.725亿美元。
国际市场上,本周共签署了80项资产授权和合作协议。最大的一项交易是Astellas与Vir Biotechnology达成战略合作,联合开发临床1/2期资产VIR-5500,首付款2.4亿美元,总金额可达16.3亿美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China IL |
Biomica | Lishan Biomedicine 力山生物 |
BMC128 | renal cell carcinoma (RCC); non-small cell lung cancer (NSCLC); melanoma | Phase I | Undisclosed | Undisclosed | Global |
| China IL |
Hanmi Pharmaceuticals | Zhongfu Pharmaceutical 众弗医药 |
amlodipine + losartan | aortic valve insufficiency; hypertension | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL |
Teikoku Pharma USA | China Medical System 康哲药业 |
lidocaine | postherpetic neuralgia (PHN); local anesthesia; dermal filler injection; pulsed dye laser therapy; facial laser resurfacing; laser-assisted tattoo removal | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL |
Formycon | Lotus Pharmaceutical 美时化学制药 |
pembrolizumab biosimilar | melanoma; non-squamous non-small cell lung cancer | Phase III | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan; Japan; Other |
| China IL |
Almirall | Sinomune Pharmaceutical 知原药业 |
sarecycline | acne; acne vulgaris; ischemic stroke | Approved | Undisclosed | Undisclosed | Chinese mainland; Other |
| China OL |
Innovent Biologics 信达生物 |
Eli Lilly | Innovative drug research and development | oncology; immunology | Pre-Clinical | 8,850 | 350 | United States; Europe; Japan; Other |
| China OL |
Sanegene Bio 圣因生物 |
Genentech | RNAi therapy | unknown/to be determined | Pre-Clinical | 1,500 | 200 | Global |
| China OL |
GenHouse 勤浩医药 |
Gilead Sciences | GH31 | oncology; solid tumor | Phase I | 1,530 | 80 | Global |
| China OL |
Henlius Biotech 复宏汉霖 |
Eisai | serplulimab | Gastroenterology; Infection; Rare Disease; Hematology; Immunology; Oncology | Approved | 388 | 75 | Japan |
| China OL |
Ribo Life Science 瑞博生物 |
Madrigal Pharmaceuticals | Six siRNA therapies targeting MASH | metabolic dysfunction-associated steatohepatitis (MASH) | Pre-Clinical | 4,460 | 60 | Global |
| China OL |
Harbour BioMed 和铂医药 |
Solstice Oncology | porustobart | NET; solid tumor; NSCLC; melanoma; RCC; HCC | Phase I/II | 1,155 | 50 | United States; Europe; Japan; Other |
| China OL |
Frontier Biotechnologies 前沿生物 |
GSK | Two small nucleic acid (siRNA) pipelines | Undisclosed | Pre-Clinical | 1,003 | 40 | Global |
| China OL |
Bio-Thera Solutions 百奥泰 |
Avalon Pharma | pembrolizumab biosimilar | solid tumor; NSCLC | Phase III | 72 | Undisclosed | Other |
| China OL |
Chipscreen Biosciences 微芯生物 |
Nizhpharm | chidamide | Infection; Hematology; Rare Disease; Immunology; Oncology | Approved | Undisclosed | 1 | Other |
| China OL |
WuXi XDC 药明合联 |
Earendil Labs | WuXiTecan-2 payload-linker technology platform; next-generation ADCs | Undisclosed | Pre-Clinical | 885 | Undisclosed | Global |
| China OL |
Sciwind Biosciences 先为达生物 |
Pfizer | ecnoglutide | MASH; obesity; hypertension; type 2 diabetes; diabetic nephropathy; dyslipidemia; overweight; OSA | Approved | 495 | Undisclosed | Chinese mainland |
| China OL |
Dartsbio Pharmaceuticals 达石药业 |
Slate Medicines | DS009 | migraine; headache | Pre-Clinical | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| China OL |
Formosa Pharmaceuticals 台新药 |
Arrotex | APP13007 (clobetasol propionate nanoparticle) | cataract; ocular postoperative inflammation; ocular postoperative pain | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Jingxin Pharmaceutical 京新药业 |
AMB HK Enterprises | dimdazenil | insomnia; excessive daytime sleepiness (EDS) | Approved | Undisclosed | Undisclosed | Other |
| China OL |
WuXi Biologics 药明生物 |
Vertex Pharmaceuticals | Trispecific T cell engager | autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China OL |
Zonsen Biotech 中晟全肽 |
Eli Lilly | new peptide drugs | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
Micot Technology 麦科奥特 |
Everest Medicines 云顶新耀 |
MT1013 | secondary hyperparathyroidism in CKD; HPT; CKD | Phase III | 172.5 | 28 | Chinese mainland; Hong Kong, Macao and Taiwan; Other |
| China Domestic |
Akeso Biopharma 康方生物 |
Jumpcan Pharmaceutical 济川药业 |
ebronucimab | mixed dyslipidemia; cardiovascular risk; HoFH; High LDL Cholesterol; HeFH; hypercholesterolemia; dyslipidemia | Approved | 12.5 | 11 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China Domestic |
Thederma 泽德曼 |
Jumpcan Pharmaceutical 济川药业 |
TAP-1503 (1% benvitimod cream) | alopecia areata; eczema; vitiligo; atopic dermatitis | Approved | 26 | Undisclosed | Chinese mainland |
| China Domestic |
Huisheng Pharm 惠升生物 |
Tonghua Dongbao Pharmaceutical 通化东宝 |
insulin degludec biosimilar; insulin aspart + insulin degludec | diabetes; type 2 diabetes | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic |
YolTech Therapeutics 尧唐生物 |
XtalPi 晶泰科技 |
CAR-T therapy; mRNA drugs | solid tumor; disorder of nervous system | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
LexBio 朗睿生物 |
Hiteck Biological Pharma 海特生物 |
novel drug candidates | inflammatory | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
SJTU 上海交通大学 |
Shanghai Pharmaceuticals 上海医药 |
MCB-D166 | depressive disorder | Undisclosed | Undisclosed | Undisclosed | Global |
| China Domestic |
XtalPi 晶泰科技 |
VISEN Pharmaceuticals 维昇药业 |
Candidate drugs based on the AI platform | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
Vcare PharmaTech 威凯尔 |
SunNovo 阳光诺和 |
VC005 | IBD; alopecia areata; vitiligo; eczema; atopic dermatitis; RA; AS | Phase III | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic |
Insilico Medicine 英矽智能 |
China Medical System 康哲药业 |
Pharma.AI candidates | disorder of nervous system; autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
Milelonge Pharma (Huading Modern Bio-pharmaceutical) 迈乐生物 |
Main Luck Pharmaceuticals 万乐药业 |
hematoporphyrin | oral cancer; bladder cancer; lung cancer; gastrointestinal cancer; skin cancer | Approved | Undisclosed | Undisclosed | Chinese mainland |
Global Licensing and Cooperation Deals February 2026
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License; Investment | Vir Biotechnology | Astellas Pharma | AMX-500 | castration-resistant prostate cancer (CRPC) | Phase I/II | 1,630 | 240 | Global |
| Cooperation; License | Unnatural Products | Novartis | macrocyclic peptides | cardiovascular disease | Pre-Clinical | 1,800 | 100 | Global |
| License | CSL | Eli Lilly | clazakizumab | Respiratory; Gastroenterology; Infection; Rare Disease; Musculoskeletal; Immunology; Cardiovascular; Oncology; Genitourinary; Endocrine & Metabolic | Phase III | Undisclosed | 100 | Global |
| License | THX Pharma | Laboratoires Biocodex | TX01 (miglustat oral liquid formulation); Batten-1 (miglustat) | Gaucher’s disease type I; Niemann-Pick disease type C; neuronal ceroid lipofuscinosis (NCL); CLN3 disease | Pre-Clinical; Phase I/II | 196 | 14 | Global; United States; Other |
| License; Investment | Invenra | Twist Bioscience | B-Body® bispecific antibody platform | Undisclosed | Undisclosed | Undisclosed | 5 | Global |
| License | Theriva Biologics | Rasayana Therapeutics | SYN-020 | metabolic disease; acute radiation enteritis | Phase I | 38 | 0.30 | Global |
| Cooperation; License | Vivtex | Novo Nordisk | next-generation oral biologic medicines | obesity; diabetes | Pre-Clinical | 2,100 | Undisclosed | Global |
| License | Araris Biotech | Chugai Pharmaceutical | AraLinQ ADC; aralinq® linker-payload platform | cancer | Pre-Clinical | 780 | Undisclosed | Global |
| Option; License | Memo Therapeutics | CSL | recombinant polyclonal IgG products | Undisclosed | Pre-Clinical | 328 | Undisclosed | Global |
| Cooperation | Quiver Biosciences | Angelini | novel therapeutics | developmental and epileptic encephalopathy | Pre-Clinical | 120 | Undisclosed | Global |
| License | Sam Chun Dang | Undisclosed | semaglutide biosimilar | type 2 diabetes | Phase I | 32 | Undisclosed | Europe |
| Cooperation | Eckert & Ziegler | Molecular Partners | Radio-DARPin therapeutics | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Celyn Therapeutics | Kairos Pharma | CL-741; CL-273 | non-small cell lung cancer (NSCLC) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Divestiture | Medimetriks Pharmaceuticals | Acrotech | difamilast | atopic dermatitis | Approved | Undisclosed | Undisclosed | United States; Other |
| Cooperation | Eckert & Ziegler | Molecular Partners | Radio-DARPins | oncology | Undisclosed | Undisclosed | Undisclosed | Global |
| License | Sitryx | Boehringer Ingelheim | preclinical, small molecule program | inflammatory (not otherwise specified); autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | George Health | Orient Europharma | GMRx2 | hypercholesterolemia; hypertension; ischemic stroke; transient ischemic attack (TIA) | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | InVirtuoLabs | Helsinn Healthcare | novel drug candidates | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Shilpa Biologicals | SteinCares | a biosimilar | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Other |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Asset Acquisition | Charles River Lab | IQVIA | certain discovery services assets | oncology; neurology; immunology; metabolic disease; rare disease | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Sanacor | Chemify | oral small-molecule inhibitors | type 2 diabetes | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Grünenthal | Clinect | Qutenza (capsaicin) | Neurology; Anesthesia & Analgesia; Musculoskeletal; Rare Disease; Oncology | Approved | Undisclosed | Undisclosed | Other |
| License | NovaRock Biotherapeutics | LCB | Novel anti-tumor targeted antibodies | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Duke University | Avenue Therapeutics | clenbuterol | asthma; chronic obstructive pulmonary disease (COPD) | Approved | Undisclosed | Undisclosed | Global |
| Cooperation | Sylamore Bio | Daiichi Sankyo | SyLEC™ delivery technology | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Rznomics | Therapeutics | AAV-mediated RNA gene therapy | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Solaxa | Alvogen | SLX-100 | spinocerebellar ataxia | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation | ImmunityBio | Cigalah Healthcare; Biopharma-mea | nogapendekin alfa inbakicept-pmln | Infection; Hematology; Rare Disease; Immunology; Oncology | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | Ehime University | Sumitomo Pharma | multi-stage malaria vaccine | malaria | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | ImmunityBio | Accord Healthcare | nogapendekin alfa inbakicept-pmln | Infection; Hematology; Rare Disease; Immunology; Oncology | Approved | Undisclosed | Undisclosed | Europe; Other |
| Cooperation | Outlook Therapeutics | Mediconsult AG | bevacizumab biosimilar | Ophthalmology; Oncology | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | Asahi Kasei Pharma | Maia Pharmaceuticals | Teribone (once-weekly teriparatide) | osteoporosis | Approved | Undisclosed | Undisclosed | United States |
| License | Apertura Gene Therapy | Finding Hope for Frizzle | TfR1 CapX technology | disorder of nervous system | Undisclosed | Undisclosed | Undisclosed | Global |
| License | McMaster University | Stoked Bio | enterololin | Crohn’s disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Asset Acquisition | BriaCell Therapeutics | BriaPro Therapeutics | sCD80 program | solid tumor | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | S.Biomedics | Catalent | TED-A9 | Parkinson’s disease (PD) | Phase I/II | Undisclosed | Undisclosed | Global |
| Cooperation; License; Option | FimmCyte | Richter Group | FMC2 | endometriosis | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Mayo Clinic | Merck & Co. | drug discovery and development | inflammatory bowel disease (IBD); atopic dermatitis; multiple sclerosis (MS) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Emory University | GeoVax Labs | Ad/PNP; Gedeptin® in combination with immune checkpoint inhibitors | head and neck cancer solid tumor | Phase I/II | Undisclosed | Undisclosed | Global |
| Cooperation; License | GelMEDIX | Undisclosed | hydrogel scaffold technology | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Bayer | Essential Pharma Group | iloprost | Respiratory; Infection; Musculoskeletal; Rare Disease; Cardiovascular; Immunology; Dermatology; Oncology; Other; Genitourinary | Approved | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan; Europe; Japan; Other |
| Cooperation | Teijin | ASKA Pharmaceutical | gynecological diseases candidates | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Alchemedicine | Asahi Kasei Pharma | HiSAP lead compounds | autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Evogene | Queensland University of Technology | novel small molecules | non-small cell lung cancer (NSCLC) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Nektar Therapeutics | University of California, San Francisco | NKTR-0165 | autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | FRONTEO | Maruishi Pharmaceutical | drug discovery | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Lilac Biosciences | SNI | RNA Signatures in Pain and Neuromodulation | pain | Undisclosed | Undisclosed | Undisclosed | Global |
| License | Zifo | Maze Therapeutics | precision medicine | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | Helsinn Healthcare | Esteve | netupitant+palonosetron; palonosetron | Gastroenterology; Rare Disease; Oncology | Approved | Undisclosed | Undisclosed | Other |
| License | Nxera Pharma | Undisclosed | GPCR-targeted project | Undisclosed | Undisclosed | Undisclosed | Undisclosed | United States; Europe; Other |
| Cooperation | NBHL | FRONTEO | NBHL’s existing pipeline; new antibody drug pipeline | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Zelluna | Medpace | ZI-MA4-1 | sarcoma; gastric cancer; ovarian cancer (OVC); head and neck cancer; melanoma; solid tumor; non-small cell lung cancer (NSCLC) | IND | Undisclosed | Undisclosed | Global |
| License | Algok Bio | Keeps Biopharma | idetrexed | solid tumor; ovarian cancer (OVC) | Phase I | Undisclosed | Undisclosed | Other |
| Cooperation | Niowave | Novartis | actinium-225 | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| License | NovaCell Technology | Dongkoo Bio&Pharma | NCP112 | atopic dermatitis; ophthalmology; dry eye syndrome (DES) | Phase I | Undisclosed | Undisclosed | Global |
| Cooperation | Citius Oncology | Uniphar | denileukin diftitox | Rare Disease; Hematology; Immunology; Oncology | Approved | Undisclosed | Undisclosed | Europe |
| Cooperation; Option; License | Key2brain | OliX Pharmaceuticals | Key2Brain’s BBB shuttle technology; siRNA-BBB shuttle conjugate | disorder of nervous system | PreClinical | Undisclosed | Undisclosed | Global |
| Cooperation | AbClon | Strike Pharma | STR3/19 | autoimmune disease | PreClinical | Undisclosed | Undisclosed | Global |
| Cooperation | Helsinn Healthcare | MagnaPharm | palonosetron; netupitant+palonosetron | chemotherapy induced nausea and vomiting (CINV); PONV; peritoneal cancer; fallopian tube cancer; postoperative nausea and vomiting (PONV); nausea and vomiting; ovarian cancer (OVC); chemotherapy induced nausea and vomiting (CINV) | Approved | Undisclosed | Undisclosed | Europe |
| Cooperation | Ethris | DZIF | mRNA-based vaccines | infection; virus infection; bacterial infection; parasitic disease | PreClinical | Undisclosed | Undisclosed | Global |
| Cooperation | Galux | Isu Abxis | Rare disease treatment drugs | rare disease | PreClinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Iambic Therapeutics | Takeda Pharmaceuticals | high-priority small molecule programs | oncology; gastrointestinal disease; inflammatory | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Tohoku University | Pharma Foods | CasMab antibody | cancer | PreClinical | Undisclosed | Undisclosed | Global |
| Cooperation | Clearmind Medicine | Polyrizon | Chaperon | binge eating disorder (BED); cocaine addiction; alcohol dependence; obesity | Phase I/II | Undisclosed | Undisclosed | Global |
| Cooperation | GlyTech | FRONTEO | GT-02037 | acromegaly | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation | JURA Bio | Koa Bio | multidrug-resistant antibacterial drugs | bacterial infection | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Grünenthal | Apotex | testosterone undecanoate | androgen deficiency; hypogonadism | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | OPKO Biologics | Entera Bio | LA-PTH hypoparathyroidism program | hypoparathyroidism | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Otsuka Pharmaceutical | Biodexa Pharmaceuticals | OPB-171775 | gastrointestinal stromal tumor (GIST); solid tumor | Phase I | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan; United States; Europe; Other |
| Asset Acquisition | Hookipa Pharma | NeoTrail Therapeutics | HB-700; eseba-vec | cancer HPV-related cancer; head and neck cancer; oropharyngeal cancer; heartburn; squamous cell carcinoma of the head and neck (SCCHN) | IND; Phase II/III | Undisclosed | Undisclosed | Global |
| License | Gates MRI | SK Bioscience | RSM01 | RSV infection | Phase I | Undisclosed | Undisclosed | Other; Global |
| Cooperation | Merck & Co. | KwangDong Pharmaceutical | V116 | S. pneumoniae infection | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | Vect-Horus | OliX Pharmaceuticals | siRNA delivery research targeting central nervous system (CNS) disorders | disorder of nervous system | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Mujinmedi | Chong Kun Dang Pharmaceutical | AD-303 (dutasteride) | androgenic alopecia (AGA) | Phase I/II | Undisclosed | Undisclosed | Global |
| Cooperation | Madrigal Pharmaceuticals | Swixx | resmetirom | hepatic fibrosis; heterozygous familial hypercholesterolemia (HeFH); metabolic dysfunction associated fatty liver disease (MAFLD); liver cirrhosis; metabolic dysfunction-associated steatohepatitis (MASH) | Approved | Undisclosed | Undisclosed | Europe; Other |
| License | ZipBio | MeiraGTx | targeting complement pathway therapy | geographic atrophy | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Novartis | Senju Pharmaceutical | brolucizumab | macular edema; diabetic macular edema (DME); wet age-related macular degeneration; macular edema following retinal vein occlusion (MEfRVO); choroidal neovascularisation (CNV); age-related macular degeneration (AMD); retinal vein occlusion (RVO); polypoidal choroidal vasculopathy (PCV); proliferative diabetic retinopathy | Approved | Undisclosed | Undisclosed | Japan |
| Cooperation | 48Hour Discovery | AdvanCell | 212Pb radioligand therapy | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Alvotech | Sandoz | biosimilar candidate in ophthalmology; three biosimilar candidates across immunology and gastroenterology | ophthalmology immunology; gastrointestinal disease | Undisclosed | Undisclosed | Undisclosed | Other |
| License; Divestiture | Gustave Roussy; SATT Paris-Saclay | Kahimmune Therapeutics | Kahigens™ neoantigens; Kahinomics™ platform; shared mRNA cancer vaccines | cancer colorectal cancer (CRC); pancreatic cancer; cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
M&A Deals February 2026
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) | Upfront ($ million) |
|---|---|---|---|---|---|---|
| Proposed | AiCuris Anti-infective Cures | Veloxis Pharmaceuticals | Full Acquisition | Letermovir; pritelivir; AIC468 | 882 | 882 |
| Proposed | Arcellx | Gilead Sciences | Full Acquisition | anitocabtagene autoleucel | 7800 | Undisclosed |
| Proposed | Orna Therapeutics | Eli Lilly | Full Acquisition | CD19 panCAR therapy | 2400 | Undisclosed |
| Proposed | 35Pharma | GSK | Full Acquisition | HS235 | 950 | Undisclosed |
| Proposed | AiCuris Anti-infective Cures | Asahi Kasei | Full Acquisition | Letermovir; AIC468; pritelivir | 882 | Undisclosed |
| Completed | Faeth Therapeutics | Sensei Biotherapeutics | Full Acquisition | Undisclosed | Undisclosed | Undisclosed |
| Proposed | Novakand | SVF Vaccines | Reverse Takeover | SVF-001 | Undisclosed | Undisclosed |
2a. In-Licensing Deals
2b. Out-Licensing Deals


2c. Domestic Licensing Deals

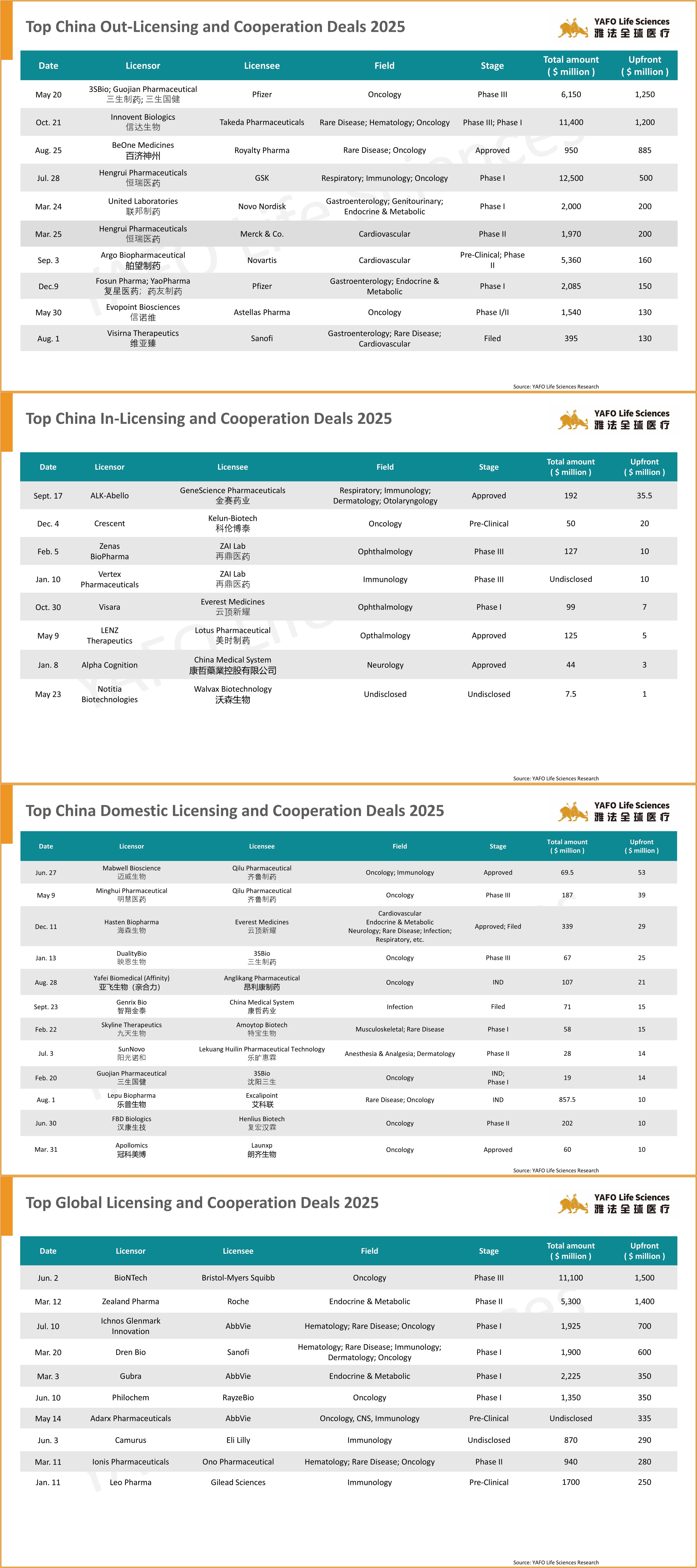
3. Top Deals of the Year 2025
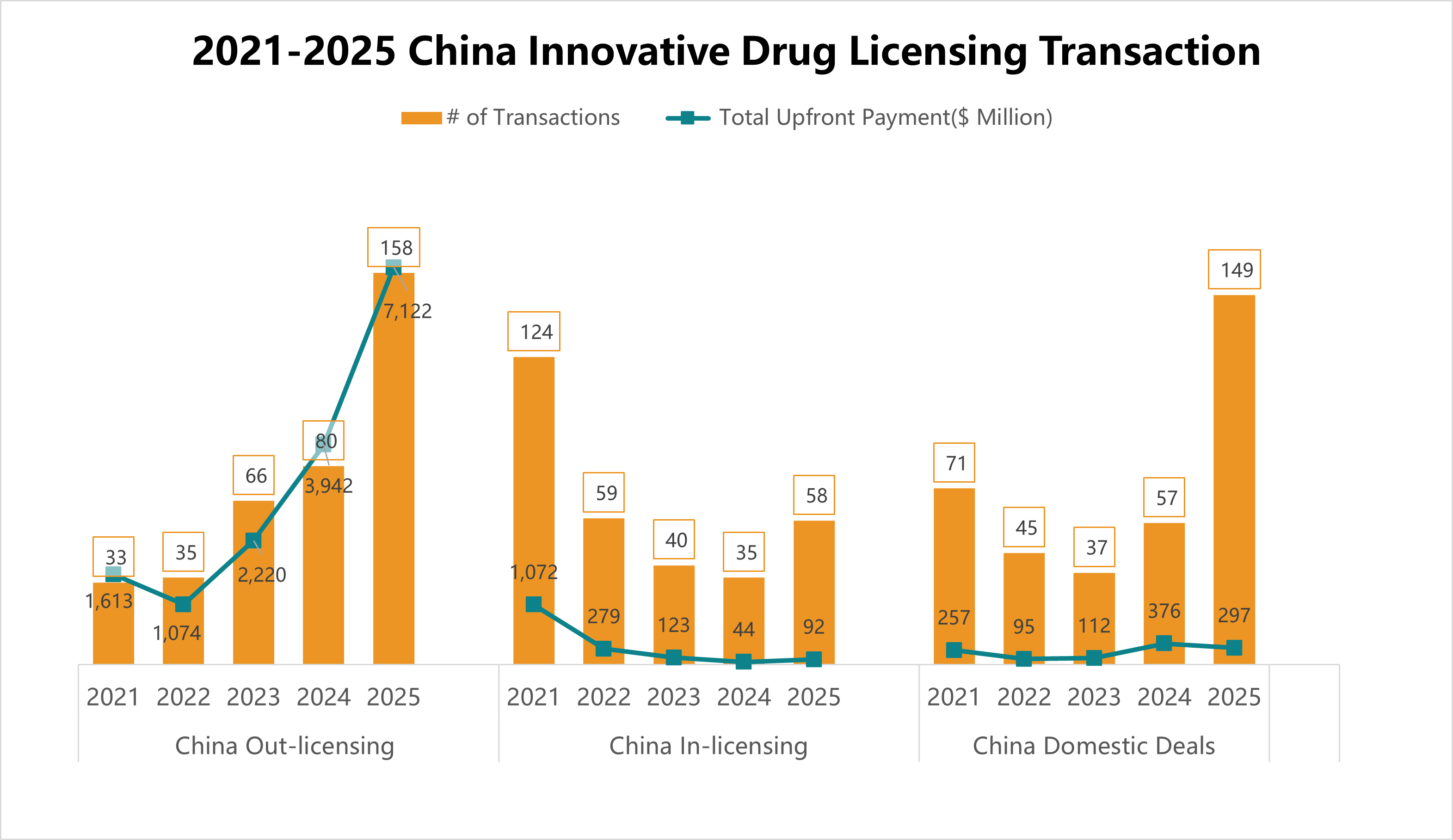
4. 2021-2025 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。