1. Summary of the Week
From March 14 to 20, a total of 19 agreements were signed worldwide. Within China’s biotech industry, there were three out-licensing deals and three domestic deals.
The top out-licensing deal in China was Alebund, granting R1 Therapeutics exclusive rights outside Greater China for the Phase 2 asset AP306, with potential milestone payments of up to low triple-digit millions of U.S. dollars, tiered royalties, and a substantial equity interest in R1.
Globally, 13 deals were announced. The top deal was between Synnovation Therapeutics and Novartis for the Phase I/II asset SNV4818 and other PI3Kα inhibitor programs, with an upfront payment of $2 billion and a total deal value of $3 billion.
2026年3月14日-20日,全球医药市场共签署了19项资产授权和合作协议。中国市场共达成6项交易,包括3项出海交易和3项国内交易。
国内市场上,本周最大的出海交易是礼邦医药授予R1 Therapeutics二期资产AP306在大中华区以外地区的独家权利,礼邦医药将获得最高总计过亿美元的里程碑付款、特许权使用费、以及R1 Therapeutics的股权。 国际市场上,本周共签署了13项资产授权和合作协议。最大的交易是Novartis从Synnovation Therapeutics收购包括临床½期资产SNV4818在内的一系列泛突变选择性PI3Kα抑制剂项目组合,首付款20亿美元,总金额可达30亿美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China OL |
Medigen Vaccine 高端疫苗生物制剂 |
Substipharm | Envacgen | enterovirus infection; virus infection; infection; hand, foot and mouth disease (HFMD) | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Alebund Pharmaceuticals 礼邦医药 |
R1 therapeutics | AP306 | hyperphosphatemia in chronic kidney disease | Phase II | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| China OL |
Auson Pharma 奥全生物 |
Undisclosed | AUSUSVAR (Rivaroxaban dispersible tablet) | deep vein thrombosis (DVT); pulmonary embolism (PE) | Approved | Undisclosed | Undisclosed | United States |
| China Domestic |
PegBio 派格生物 |
Tenry Pharma 腾瑞制药 |
visepegenatide | diabetes; overweight; chronic renal insufficiency; diabetic cardiovascular disease; type 2 diabetes; Alzheimer’s disease (AD); obesity; cardiovascular risk | Approved | 18 | Undisclosed | Chinese mainland |
| China Domestic |
Kintor Pharmaceuticals 开拓药业 |
Guangzhou Manner; Shanghai Manner Chemicals 万豪化工; 方登化工 |
KT-939 | hyperpigmentation | Pre-Clinical | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic |
Ab&B Bio-Tech 中慧元通 |
Walvax Biotechnology 沃森生物 |
Influenza virus subunit vaccines | influenza | Approved | Undisclosed | Undisclosed | Chinese mainland; United States; Europe; Japan; Other |
2a. China Section

2b. Global Section
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Asset Acquisition | Synnovation Therapeutics | Novartis | SNV4818; other PI3Kα inhibitor programs | breast cancer; cancer; solid tumor | Phase I/II | 3,000 | 2,000 | Global |
| Asset Acquisition | Corium Therapeutics | Collegium Pharmaceutical | serdexmethylphenidate + dexmethylphenidate | attention deficit hyperactivity disorder (ADHD) | Approved | 785 | 650 | United States |
| Asset Acquisition | Zevra Therapeutics | Commave Therapeutics | serdexmethylphenidate + dexmethylphenidate; serdexmethylphenidate | attention deficit hyperactivity disorder (ADHD); narcolepsy; idiopathic hypersomnia; drug dependence; attention deficit hyperactivity disorder (ADHD) | Approved; Phase III | 50 | 50 | Global |
| Cooperation | Leveragen | Daiichi Sankyo | advanced biologics | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Zydus Lifesciences | Torrent Pharmaceuticals | semaglutide biosimilar | obesity; type 2 diabetes | Phase III | Undisclosed | Undisclosed | Other |
| License | Valneva | Elaris | VLA84 | Clostridium difficile infection (CDI) | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation | Zydus Lifesciences | Lupin | semaglutide biosimilar | obesity; type 2 diabetes | Phase III | Undisclosed | Undisclosed | Other |
| Cooperation | Alloy Therapeutics | AbbVie | antibody discovery platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Voro Therapeutics | Daiichi Sankyo | Tumor-Activated ADCs | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | George Health | Arrotex | GMRx2 | hypercholesterolemia; hypertension; ischemic stroke; TIA | Approved | Undisclosed | Undisclosed | Other |
| Asset Acquisition | Genexine | Egret Therapeutics | GX-P1 | autoimmune disease; SAH | Phase I | Undisclosed | Undisclosed | Global; Other |
| Asset Acquisition | PRG S&Tech | Sentyln Therapeutics | progerinin | Werner syndrome; HGPS | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation; License; Option | G2GBio | Samsung Bioepis; Ep Epis NexLab | GB-7001; long-acting microsphere drug delivery platform | diabetes | Pre-Clinical | Undisclosed | Undisclosed | Global |
3. M&A Deals
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| Completed | Radius Health | Paratek Pharmaceuticals | Merge | abaloparatide | Undisclosed | Undisclosed |
4. Top Deals of 2025
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| May 20 | 3SBio; Guojian Pharmaceutical 三生制药; 三生国健 |
Pfizer | Oncology | Phase III | 6,150 | 1,250 |
| Oct. 21 | Innovent Biologics 信达生物 |
Takeda Pharmaceuticals | Rare Disease; Hematology; Oncology | Phase III; Phase I | 11,400 | 1,200 |
| Aug. 25 | BeOne Medicines 百济神州 |
Royalty Pharma | Rare Disease; Oncology | Approved | 950 | 885 |
| Jul. 28 | Hengrui Pharmaceuticals 恒瑞医药 |
GSK | Respiratory; Immunology; Oncology | Phase I | 12,500 | 500 |
| Mar. 24 | United Laboratories 联邦制药 |
Novo Nordisk | Gastroenterology; Genitourinary; Endocrine & Metabolic | Phase I | 2,000 | 200 |
| Mar. 25 | Hengrui Pharmaceuticals 恒瑞医药 |
Merck & Co. | Cardiovascular | Phase II | 1,970 | 200 |
| Sep. 3 | Argo Biopharmaceutical 朗誉制药 |
Novartis | Cardiovascular | Pre-Clinical; Phase II | 5,360 | 160 |
| Dec.9 | Fosun Pharma; YaoPharma 复星医药; 药友制药 |
Pfizer | Gastroenterology; Endocrine & Metabolic | Phase I | 2,085 | 150 |
| May 30 | Evopoint Biosciences 信诺维 |
Astellas Pharma | Oncology | Phase I/II | 1,540 | 130 |
| Aug. 1 | Visirna Therapeutics 维亚臻 |
Sanofi | Gastroenterology; Rare Disease; Cardiovascular | Filed | 395 | 130 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| Sept. 17 | ALK-Abello | GeneScience Pharmaceuticals 金赛药业 |
Respiratory; Immunology; Dermatology; Otorhinolaryngology | Approved | 192 | 35.5 |
| Dec. 4 | Crescent | Kelun-Biotech 科伦博泰 |
Oncology | Pre-Clinical | 50 | 20 |
| Feb. 5 | Zenas BioPharma | ZAI Lab 再鼎医药 |
Ophthalmology | Phase III | 127 | 10 |
| Jan. 10 | Vertex Pharmaceuticals | ZAI Lab 再鼎医药 |
Immunology | Phase III | Undisclosed | 10 |
| Oct. 30 | Visara | Everest Medicines 云顶新耀 |
Ophthalmology | Phase I | 99 | 7 |
| May 9 | LENZ Therapeutics | Lotus Pharmaceutical 美时制药 |
Ophthalmology | Approved | 125 | 5 |
| Jan. 8 | Alpha Cognition | China Medical System 康哲药业控股有限公司 |
Neurology | Approved | 44 | 3 |
| May 23 | Notitia Biotechnologies | Walvax Biotechnology 沃森生物 |
Undisclosed | Undisclosed | 7.5 | 1 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| Jun. 27 | Mabwell Bioscience 迈威生物 |
Qilu Pharmaceutical 齐鲁制药 |
Oncology; Immunology | Approved | 69.5 | 53 |
| May 9 | Minghui Pharmaceutical 明慧医药 |
Qilu Pharmaceutical 齐鲁制药 |
Oncology | Phase III | 187 | 39 |
| Dec. 11 | Hasten Biopharma 海森生物 |
Everest Medicines 云顶新耀 |
Cardiovascular Endocrine & Metabolic Neurology; Rare Disease; Infection; Respiratory, etc. |
Approved; Filed | 339 | 29 |
| Jan. 13 | DualityBio 映恩生物 |
3SBio 三生制药 |
Oncology | Phase III | 67 | 25 |
| Aug. 28 | Yafei Biomedical (Affinity) 亚飞生物(亲合力) |
Anglikang Pharmaceutical 昂利康制药 |
Oncology | IND | 107 | 21 |
| Sept. 23 | Genrix Bio 智翔金泰 |
China Medical System 康哲药业 |
Infection | Filed | 71 | 15 |
| Feb. 22 | Skyline Therapeutics 九天生物 |
Amytop Biotech 诗至生物 |
Musculoskeletal; Rare Disease | Phase I | 58 | 15 |
| Jul. 3 | SunNovo 阳光诺和 |
Lekuang Huilin Pharmaceutical Technology 乐旷惠霖 |
Anesthesia & Analgesia; Dermatology | Phase II | 28 | 14 |
| Feb. 20 | Guojian Pharmaceutical 三生国健 |
3SBio 沈阳三生 |
Oncology | IND; Phase I |
19 | 14 |
| Aug. 1 | Leap Biopharma 乐普生物 |
Excalipoint 艾科联 |
Rare Disease; Oncology | IND | 857.5 | 10 |
| Jun. 30 | FBD Biologics 汉康生技 |
Henlius Biotech 复宏汉霖 |
Oncology | Phase II | 202 | 10 |
| Mar. 31 | Apollomics 冠科美博 |
Luanxo 朗齐生物 |
Oncology | Approved | 60 | 10 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| Jun. 2 | BioNTech | Bristol-Myers Squibb | Oncology | Phase III | 11,100 | 1,500 |
| Mar. 12 | Zealand Pharma | Roche | Endocrine & Metabolic | Phase II | 5,300 | 1,400 |
| Jul. 10 | Ichnos Glenmark Innovation |
AbbVie | Hematology; Rare Disease; Oncology | Phase I | 1,925 | 700 |
| Mar. 20 | Dren Bio | Sanofi | Hematology; Rare Disease; Immunology; Dermatology; Oncology | Phase I | 1,900 | 600 |
| Mar. 3 | Gubra | AbbVie | Endocrine & Metabolic | Phase I | 2,225 | 350 |
| Jun. 10 | Philochem | RayzeBio | Oncology | Phase I | 1,350 | 350 |
| May 14 | Adarx Pharmaceuticals | AbbVie | Oncology, CNS, Immunology | Pre-Clinical | Undisclosed | 335 |
| Jun. 3 | Camurus | Eli Lilly | Immunology | Undisclosed | 870 | 290 |
| Mar. 11 | Ionis Pharmaceuticals | Ono Pharmaceutical | Hematology; Rare Disease; Oncology | Phase II | 940 | 280 |
| Jan. 11 | Leo Pharma | Gilead Sciences | Immunology | Pre-Clinical | 1700 | 250 |
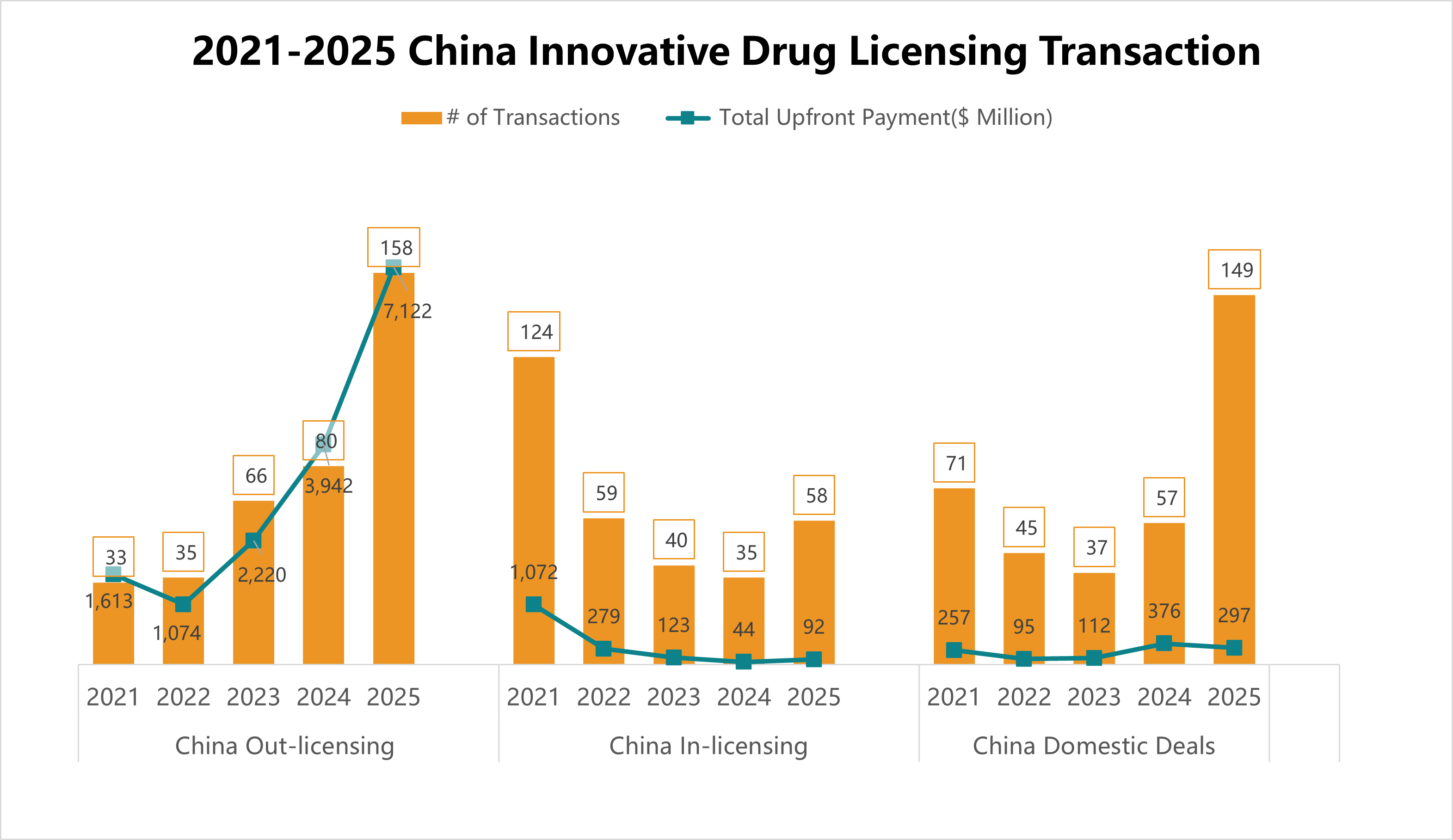
5. 2019-2024 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。