1. Executive Summary of the Month
In March 2026, 112 biopharmaceutical agreements were announced worldwide. In China’s biotech sector, there were 15 out-licensing deals, 6 in-licensing deals, and 14 domestic partnerships.
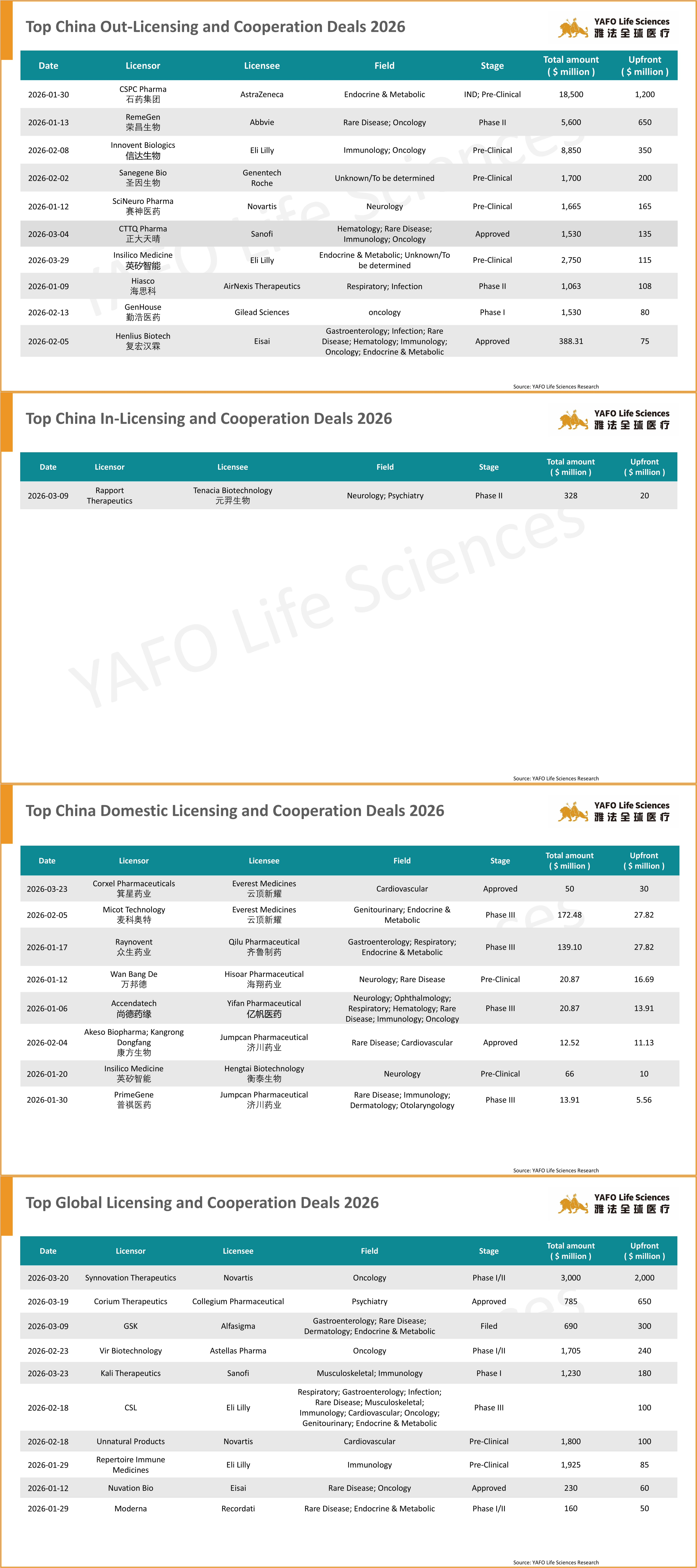
In China, the largest in-licensing deal involved Rapport Therapeutics and Tenacia Biotechnology for the Phase II asset RAP-219, featuring an upfront payment of $20 million and a total deal value of $328 million. The largest out-licensing deal was signed by Chia Tai Tianqing Pharmaceutical and Sanofi for the approved asset rovadicitinib, with an upfront payment of $135 million and a total deal value of $1.53 billion. The largest domestic deal was between Corxel Pharmaceuticals and Everest Medicines for the approved asset etripamil, including an upfront payment of $30 million and a total deal value of $50 million.
Internationally, 77 cross-border deals were reported, with the largest being the agreement between SynnovationTherapeutics and Novartis for the Phase I/II asset SNV4818 and other PI3Kα inhibitor programs, with an upfront payment of $2 billion and a total deal value of $3 billion.
2026年3月,全球医药市场共签署了112项资产授权和合作协议。中国市场共达成35项交易,包括15项出海交易,6项引进交易和14项国内交易。
国内市场上,本月最大的引进交易是元羿生物获得Rapport Therapeutics临床二期资产RAP-219的大中华区独家权益,首付款2000万美元,总金额可达3.28亿美元;本月最大的出海交易是中生制药旗下正大天晴药业与Sanofi就已获批资产rovadicitinib达成全球授权许可协议,首付款1.35亿美元,总金额可达15.3亿美元。本月最大的国内交易是云顶新耀收购箕星药业已获批资产Etripamil的大中华区独家权益,首付款3000万美元,总金额可达5000万美元。
国际市场上,本月共签署了77项资产授权和合作协议。最大的一项交易是Novartis从SynnovationTherapeutics收购包括临床½期资产SNV4818在内的一系列泛突变选择性PI3Kα抑制剂项目组合,首付款20亿美元,总金额可达30亿美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China IL | Rapport Therapeutics | Tenacia Biotechnology 元羿生物 |
RAP-219 | epilepsy; focal onset seizure; bipolar I disorder; manic episode | Phase II | 328 | 20 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China IL | Curatis Holding | NeuPharma 润新生物 |
C-PTBE-01 | cerebral edema; brain cancer | Phase II | 101 | Undisclosed | Japan |
| China IL | Roche | CR Pharmaceutical Comm 华润医药商业 |
trastuzumab emtansine | Respiratory; Rare Disease; Hematology; Oncology | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL | Teva Pharmaceutical | Nanjing Chia-tai Tianqing Pharmaceutical 南京正大天晴 |
bendamustine | Gastroenterology; Hematology; Rare Disease; Immunology; Dermatology; Oncology; Genitourinary | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL | Eli Lilly | Zuellig Pharma 裕利医药 |
tadalafil | Cardiovascular; Genitourinary; Respiratory; Musculoskeletal; Rare Disease; Hematology | Approved | Undisclosed | Undisclosed | Other |
| China IL | GSK | Keyuan Trade 科园贸易 |
Engerix-B | hepatitis B | Approved | Undisclosed | Undisclosed | Chinese mainland |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China OL | Chia Tai Tianqing Pharmaceutical 正大天晴 |
Sanofi | rovadicitinib | Hematology; Rare Disease; Immunology; Oncology | Approved | 1,530 | 135 | Global |
| China OL | Insilico Medicine 英矽智能 |
Eli Lilly | novel oral therapeutics in preclinical development; Pharma.AI candidates | Undisclosed | Pre-Clinical | 2,750 | 115 | Global |
| China OL | Antengene 德琪医药 |
UCB | ATG-201 | B-cell malignancies; autoimmune disease | Pre-Clinical | 1,180 | 60 | Global |
| China OL | Huaota Biopharm 华奥泰生物 |
Almirall | mAb program against new target | dermatology | Pre-Clinical | 340 | Undisclosed | United States; Europe; Japan; Other |
| China OL | Antengene 德琪医药 |
Boryung Pharmaceutical | selinexor | Infection; Hematology; Rare Disease; Dermatology; Oncology | Approved | Undisclosed | Undisclosed | Other |
| China OL | Medigen Vaccine 高端疫苗生物制剂 |
Substipharm | Envacgen | enterovirus infection; virus infection; infection; hand, foot and mouth disease (HFMD) | Approved | Undisclosed | Undisclosed | Other |
| China OL | Alebund Pharmaceuticals 礼邦医药 |
R1 therapeutics | AP306 | hyperphosphatemia in chronic kidney disease | Phase II | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China OL | Bio-Thera Solutions 百奥泰 |
Intas Pharmaceuticals | golimumab biosimilar | psoriatic arthritis (PsA); ankylosing spondylitis (AS); juvenile idiopathic arthritis (JIA); axial spondyloarthritis (axSpA); ulcerative colitis (UC); rheumatoid arthritis (RA) | Approved | Undisclosed | Undisclosed | Other |
| China OL | Biocytogen 百奥赛图 |
Moonlight Bio | Cell therapy developed using the Biocytogen antibody-binding agent library | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China OL | Mabwell Bioscience 迈威生物 |
Undisclosed | denosumab biosimilar | breast cancer; bone metastasis; osteoporosis; bone cancer; giant cell tumor of bone; multiple myeloma (MM) | Approved | Undisclosed | Undisclosed | Other |
| China OL | GlycoNex 糖联生技 |
Nippon Kayaku | GNX201-ADC | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China OL | Insilico Medicine 英矽智能 |
ASKA Pharmaceutical | Women’s Health Innovative Target Discovery Project | endometriosis; uterine fibroid; uterine adenomyosis | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China OL | Auson Pharma 奥全生物 |
Undisclosed | AUSUSVAR (Rivaroxaban dispersible tablet) | deep vein thrombosis (DVT); pulmonary embolism (PE) | Approved | Undisclosed | Undisclosed | United States |
| China OL | JOYO Pharma 嘉越医药 |
Erasca | JYP0015 | colorectal cancer (CRC); non-small cell lung cancer (NSCLC); leukemia; solid tumor; pancreatic cancer; pancreatic ductal carcinoma; hematologic malignancies | Phase II | Undisclosed | Undisclosed | Global |
| China OL | Biocytogen 百奥赛图 |
Taisho Pharmaceutical | RenNano® Fully Human Heavy Chain Only Antibody Discovery Platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China Domestic | Corxel Pharmaceuticals 箕星药业 |
Everest Medicines 云顶新耀 |
etripamil | supraventricular tachycardia (SVT); paroxysmal supraventricular tachycardia (PSVT); atrial fibrillation; ventricular tachycardia (VT) | Approved | 50 | 30 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China Domestic | Insilico Medicine 英矽智能 |
Tenacia Biotechnology 元羿生物 |
Pharma.AI candidates | disorder of nervous system | Pre-Clinical | 95 | Undisclosed | Global |
| China Domestic | PegBio 派格生物 |
Tenry Pharma 腾瑞制药 |
visepegenatide | diabetes; overweight; chronic renal insufficiency; diabetic cardiovascular disease; type 2 diabetes; Alzheimer’s disease (AD); obesity; cardiovascular risk | Approved | 18 | Undisclosed | Chinese mainland |
| China Domestic | Sintanovo Biological Technology 诺和晟泰 |
Salubris Pharmaceuticals 信立泰 |
STC007 | post-operative pain; uremic pruritus | Phase II | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China Domestic | Belief BioMed 信念医药 |
Grand Life Science 远大生命科学 |
BBM-H803 | haemophilia A; haemophilia | Phase III | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan |
| China Domestic | Kintor Pharmaceuticals 开拓药业 |
Guangzhou Manner; Shanghai Manner Chemicals 万嘉化工; 万登化工 |
KT-939 | hyperpigmentation | Pre-Clinical | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic | Ab&B Bio-Tech 中慧元通 |
Walvax Biotechnology 沃森生物 |
Influenza virus subunit vaccines | influenza | Approved | Undisclosed | Undisclosed | Chinese mainland; United States; Europe; Japan; Other |
| China Domestic | Ark Biosciences 爱科百发 |
Qilu Pharmaceutical 齐鲁制药 |
serdexmethylphenidate + dexmethylphenidate | attention deficit hyperactivity disorder (ADHD) | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic | Kintor Pharmaceuticals 开拓药业 |
Fonow 孚诺医药 |
KT-939 | hyperpigmentation | Pre-Clinical | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic | Pamplona Therapeutics 潘普洛纳 |
Coherent Biopharma 同宜医药 |
New generation of innovative drugs combining degradation agents and coupling agents | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China Domestic | Westlake University 西湖大学 |
Olymvax 欧林生物 |
广谱流感疫苗 | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic | Asieris Pharmaceuticals 亚虹医药 |
Keyuan Trade 科园贸易 |
Cevira (hexaminolevulinate hydrochloride ointment photodynamic therapy system) | precancerous or dysplastic lesions caused by HPV; cervical intraepithelial neoplasia (CIN) | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic | DPTechnology 深势科技 |
Boan Biotech 博安生物 |
AI for science驱动的创新药研发 | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| China Domestic | Beijing Institute of Neurosurgery 北京神外所 |
Tasly; Beijing Rencheng Bio 天士力; 北京仁诚生物 |
dual-targeted CAR-T program | glioma | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Asset Acquisition | Synnovation Therapeutics | Novartis | SNV4818; other PI3Kα inhibitor programs | breast cancer; cancer; solid tumor | Phase I/II | 3,000 | 2,000 | Global |
| Asset Acquisition | Corium Therapeutics | Collegium Pharmaceutical | serdexmethylphenidate + dexmethylphenidate | attention deficit hyperactivity disorder (ADHD) | Approved | 785 | 650 | United States |
| License | GSK | Alfasigma | linerixibat | cholestasis; pruritis; type 2 diabetes; cholestatic pruritus; primary biliary cirrhosis (PBC) | Filed | 690 | 300 | Global |
| License | Kali Therapeutics | Sanofi | KT501 | rheumatoid arthritis (RA) | Phase I | 1,230 | 180 | Global |
| Asset Acquisition | Zevra Therapeutics | Commave Therapeutics | serdexmethylphenidate + dexmethylphenidate; serdexmethylphenidate | ADHD; narcolepsy; idiopathic hypersomnia; drug dependence | Approved; Phase III | 50 | 50 | Global |
| Cooperation | Quotient Therapeutics | Merck & Co. | novel drug targets in inflammatory bowel disease | inflammatory bowel disease (IBD) | Undisclosed | 2,200 | 20 | Global |
| License; Option | Alteogen | Biogen | ALT-B4 platform and related biologics | increasing tissue permeability | Approved; Pre-Clinical | 579 | 20 | Global |
| Cooperation | Tenaya Therapeutics | Alnylam Pharmaceuticals | novel human genetic targets | cardiovascular disease | Pre-Clinical | 1,140 | 10 | Global |
| Asset Acquisition | Poxel | Scynexis | PXL770 | AMN; diabetes; MASH; PKD | Phase II | 196 | 8 | Global |
| License | Ensol Biosciences | Leaders Cosmetics | Engedi 1000 (E1K) | osteoarthritis | Phase III | 70 | 3 | Other |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | AstraZeneca | Amani Therapeutics | clozapine + NA | schizophrenia; psychiatry | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Richter Group | Fuji Pharma | FMC2; AMH-targeting antibodies; FSH receptor agonist; JNK inhibitor | endometriosis; genitourinary disorder; endometriosis-related pain | Pre-Clinical | Undisclosed | Undisclosed | Japan; Global; Other |
| Cooperation | Flurry Powders | Experic | Spray drying inhalation and nasal drug development technology | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | NOBO Medicine | NAMU ICT | CHIP drug candidates | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Ube Industries | Kyorin Pharmaceutical | Novel drug candidates | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | RIKEN; Tokyo University of Pharmacy and Life Sciences | EpiFrontier Therapeutics | RK-701 | cancer; sickle cell disease (SCD) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License; Option; Cooperation | Oscotec | Yatiri Bio | denfivontinib | acute myeloid leukaemia (AML); solid tumor | Phase I | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Asset Acquisition | Kezar Life Sciences | Enodia Therapeutics | Sec61-based program; KZR-261; KZR-8834; KZR-540 | uveal melanoma; solid tumor; mesothelioma; colorectal cancer (CRC); castration-resistant prostate cancer (CRPC) | Pre-Clinical; Phase I | 127 | 1 | Global |
| License | KoBioLabs | Celltrion | KBL382; KC84; KBL385 | gastrointestinal disease; IBD; irritable bowel syndrome with diarrhea (IBS-D) | Pre-Clinical | 15 | 1 | Global |
| License | Biodol Therapeutics | Kyorin Pharmaceutical | BDT272 | pain | Phase I | 89 | Undisclosed | Japan; Other |
| Cooperation | MiNK Therapeutics | C-Further | CF-033 | oncology | Pre-Clinical | 1 | Undisclosed | Global |
| Cooperation | Johnson & Johnson | Kyorin Pharmaceutical | bedaquiline | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Undisclosed |
| Cooperation | Sanofi | Huons | MenQuadFi; Vaxigrip; Fluzone High-Dose; Adacel; Avaxim | meningitis; influenza; pertussis; sepsis; tetanus; diphtheria; hepatitis A | Approved | Undisclosed | Undisclosed | Undisclosed |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | Propanc Biopharma | UJA; University of Granada | anti-aging compounds | aging | Pre-Clinical | Undisclosed | Undisclosed | Europe |
| Asset Acquisition | Oramed Pharmaceuticals | Lifeward | protein oral delivery (pod™) technology | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Nosis Bio | Janssen Biotech | Connexa™ platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Apnimed | SASS; Shionogi | sultiame | insomnia; obstructive sleep apnea; epilepsy | Approved | Undisclosed | Undisclosed | Global |
| License | Genovis | Undisclosed | EndoS2 enzyme technology | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Biophytis | LynxKite | dry age-related macular degeneration candidates | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Geistlich Pharma; Panavance Therapeutics | Persevere Therapeutics | misetionamide | peritoneal mesothelioma; gynecological cancer; melanoma; ovarian cancer (OVC); pancreatic cancer; neuroendocrine tumor (NET) | Phase I/II | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | Sinopia Biosciences | Ono Pharmaceutical | undisclosed group of rare metabolic disorders | metabolic disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Investment; License | Teva Pharmaceutical | BXLS | duvakitug | ulcerative colitis (UC); Crohn’s disease; irritable bowel syndrome (IBS); asthma | Phase III | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Celmatix | Gedeon Richter | JNK inhibitor; FSH receptor agonist; anti-Müllerian hormone (AMH) targeting therapeutic antibodies | endometriosis related pain; genitourinary disorder | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Samsung Bioepis | Sandoz | vedolizumab biosimilar; other 4 biosimilar candidates | immunology | PreClinical | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| License | BioNxt Solutions | Undisclosed | BNT23001 (cladribine sublingual film) | multiple sclerosis (MS); myasthenia gravis (MG) | PreClinical | Undisclosed | Undisclosed | Other |
| License | Hopewell Therapeutics | Foxcroft Therapeutics | tissue-targeting lipid nanoparticles (ttLNPs) | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | Unleash Immuno Oncolytics | TransCode Therapeutics | UIO-525; UIO-524 | muscle-invasive bladder carcinoma (MIBC) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Seaport Therapeutics | Monash University | GlyphCele | endocrine/metabolic disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Antiverse | Cystic Fibrosis Foundation | anti-CFTR antibody | cystic fibrosis (CF) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Eli Lilly | Swixx | mirikizumab-mrkz | ulcerative colitis (UC); plaque psoriasis; Crohn’s disease; psoriasis | Approved | Undisclosed | Undisclosed | Other |
| Divestiture | Endevica | Kalohexis | A07D; mifomelatide | obesity; cachexia; pancreatic ductal carcinoma; cancer cachexia | Pre-Clinical; Phase II | Undisclosed | Undisclosed | Global |
| Cooperation; Option | Alivexis | Astellas Pharma | novel drug target molecule | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | AbClon | Chong Kun Dang Pharmaceutical | AffiMab bispecific antibodies | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Digmbio | Axceso Biopharma | CHEMDEGRADER PARP1 PROTAC | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | Pierre Fabre | Eton Pharmaceuticals | Hemangeol (propranolol) | hypertension; supraventricular arrhythmia; atrial flutter; tachycardia; hypertrophic cardiomyopathy (HCM); ventricular arrhythmia; atrial fibrillation; infantile hemangioma | Approved | Undisclosed | Undisclosed | United States |
| Option; Cooperation | Congruence Therapeutics | Ono Pharmaceutical | novel small molecule modulators | disorder of nervous system; immunology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | LGMD2L Foundation | CureRareDisease | ANO5 gene replacement therapy | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Leveragen | Daiichi Sankyo | advanced biologics | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Zydus Lifesciences | Torrent Pharmaceuticals | semaglutide biosimilar | obesity; type 2 diabetes | Phase III | Undisclosed | Undisclosed | Other |
| License | Valneva | Elaris | VLA84 | Clostridium difficile infection (CDI) | Phase II | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | PRISM BioLab | Receptor.AI | orally available small molecules | metabolic disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Aptar Group | COVIRIX | antiviral compound delivery | COVID-19; RSV infection; influenza; avian influenza | Undisclosed | Undisclosed | Undisclosed | Global |
| License | George Health | Ahn-Gook Pharmaceutical | GMRx2 (telmisartan + amlodipine + indapamide) | hypercholesterolemia; hypertension; ischemic stroke; transient ischemic attack (TIA) | Approved | Undisclosed | Undisclosed | Other |
| Cooperation; License; Option | Ouro Medicines; Gilead Sciences | Galapagos | BCMA×CD19×CD3 TCE program (gamgertamig); additional autoimmune programs | Immunology; Rare Disease; Hematology; Musculoskeletal; Dermatology; Oncology | Phase III; Pre-Clinical | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| Cooperation | Biodexa Pharmaceuticals | Tanner | eRapa (rapamycin) | familial adenomatous polyposis (FAP); non-muscle invasive bladder cancer (NMIBC); prostate cancer | Phase III | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | Toray Industries | Immunis | nalfurafine | chronic liver disease-associated pruritus; liver disease; uremic pruritus; levodopa-induced dyskinesia (LID); Parkinson’s disease (PD); anesthesia | Approved | Undisclosed | Undisclosed | United States; Europe; Other |
| Cooperation | BullFrog AI | Undisclosed | novel drug targets in MDD | major depressive disorder (MDD) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Fuji Yakuhin | Mochida Pharmaceutical | dotinurad | gout; CKD; heart failure; gouty arthritis; hyperuricemia | Approved | Undisclosed | Undisclosed | Japan |
| License | Mochida Pharmaceutical | LG Chem | dienogest | contraception; endometriosis; uterine adenomyosis | Approved | Undisclosed | Undisclosed | Other |
| Divestiture | Ono Pharmaceutical | Daito | limaprost; alprostadil | Respiratory; Musculoskeletal; Cardiovascular; Gastroenterology; Genitourinary; Ophthalmology | Approved | Undisclosed | Undisclosed | Global |
| Cooperation | KNIH | EuBiologics | Nipah virus vaccine | Nipah virus infection | Pre-Clinical | Undisclosed | Undisclosed | Other |
| License | Grünenthal | BCworld Pharm | Qutenza (capsaicin) | Neurology; Anesthesia & Analgesia; Musculoskeletal; Rare Disease; Oncology | Approved | Undisclosed | Undisclosed | Other |
| Cooperation; Option | J2H Biotech | SK Chemicals | MASH and IPF pipelines | MASH; idiopathic pulmonary fibrosis (IPF) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Children’s Mercy | Vima Therapeutics | VIM0423 | dystonia | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation | Daiichi Sankyo | Hyundai Pharmaceutical | pravastatin | HCC; CHD; hypercholesterolemia; hypertriglyceridemia; dyslipidemia; HeFH; stroke | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | Tanabe Pharma Corporation | inno.N | vadadustat | anemia; COVID-19; ESRD; CKD; ARDS | Approved | Undisclosed | Undisclosed | Other |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | Idorsia Pharma | Linkdar | daridorexant | postoperative delirium; neuroinflammation; OSA; dementia; COPD; amyloidosis; anxiety; opioid dependence; depression; Alzheimer’s disease; PTSD; insomnia | Approved | Undisclosed | Undisclosed | Other |
| License | University of Minnesota; University of Washington | CounterX Therapeutics | novel mAb and vaccine combinations | fentanyl overdose; opioid overdose | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Etcembly | Zelluna | KKLC1-targeting TCRs | oncology | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation; Investment | MAX BioPharma | Technomark LS | Oxy210 | fibrosis | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Zydus Lifesciences | Lupin | semaglutide biosimilar | obesity; type 2 diabetes | Phase III | Undisclosed | Undisclosed | Other |
| Cooperation | Alloy Therapeutics | AbbVie | antibody discovery platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Voro Therapeutics | Daiichi Sankyo | tumor-activated ADCs | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | George Health | Arrotex | GMRx2 | hypercholesterolemia; hypertension; ischemic stroke; TIA | Approved | Undisclosed | Undisclosed | Other |
| Asset Acquisition | Genexine | Egret Therapeutics | GX-P1 | autoimmune disease; SAH | Phase I | Undisclosed | Undisclosed | Global; Other |
| Asset Acquisition | PRG S&Tech | Sentynl Therapeutics | progerinin | Werner syndrome; HGPS | Phase II | Undisclosed | Undisclosed | Global |
| Cooperation; License; Option | G2GBio | Samsung Bioepis; Ep Epis NexLab | GB-7001; microsphere platform | diabetes | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) | Upfront ($ million) |
|---|---|---|---|---|---|---|
| Proposed | Apellis Pharmaceuticals | Biogen | Full Acquisition | pegcetacoplan | Undisclosed | 5,600 |
| Proposed | Ouro Medicines | Gilead Sciences | Full Acquisition | gamgertamig | 2,175 | 1,675 |
| Proposed | Transcend Therapeutics | Otsuka Pharmaceutical | Full Acquisition | methylone | 1,225 | 700 |
| Proposed | Corstasis Therapeutics | Esperion Therapeutics | Full Acquisition | RSQ-777 (bumetanide nasal spray) | Undisclosed | 75 |
| Proposed | Terns Pharmaceuticals | Merck & Co. | Full Acquisition | HS-10382 | 6,700 | Undisclosed |
| Proposed | Day One Biopharma | Servier | Full Acquisition | Undisclosed | 2,500 | Undisclosed |
| Proposed | Excellergy | Novartis | Full Acquisition | Undisclosed | 2,000 | Undisclosed |
| Proposed | Cullgen | Gyre Therapeutics | Full Acquisition | CG009301; CG001419 | 300 | Undisclosed |
| Proposed | Coiled Therapeutics | Roquefort Therapeutics | Reverse Takeover | A2A-O-0442 | Undisclosed | Undisclosed |
| Proposed | Candid Therapeutics | Rallybio | Reverse Takeover | Cizutamig; CND319; GB261 | Undisclosed | Undisclosed |
| Completed | Radius Health | Paratek Pharmaceuticals | Merge | abaloparatide | Undisclosed | Undisclosed |
| Completed | SASS | Shionogi | Full Acquisition | sivopixant + acetazolamide; sultiame | Undisclosed | Undisclosed |
| Proposed | Centessa Pharmaceuticals | Eli Lilly | Full Acquisition | ORX489; ORX142; ORX750 | Undisclosed | Undisclosed |
| Proposed | Kezar Life Sciences | Aurinia Pharmaceuticals | Full Acquisition | zetomipzomib | Undisclosed | Undisclosed |
| Completed | Lipum | Flerie | Merge | SOL-116 | Undisclosed | Undisclosed |
| Proposed | Genovate Biotechnology | Easywell Biomedicals | Full Acquisition | Undisclosed | Undisclosed | Undisclosed |
2a. In-Licensing Deals

2b. Out-Licensing Deals
2c. Domestic Licensing Deals

3. Top Deals of the Year 2026
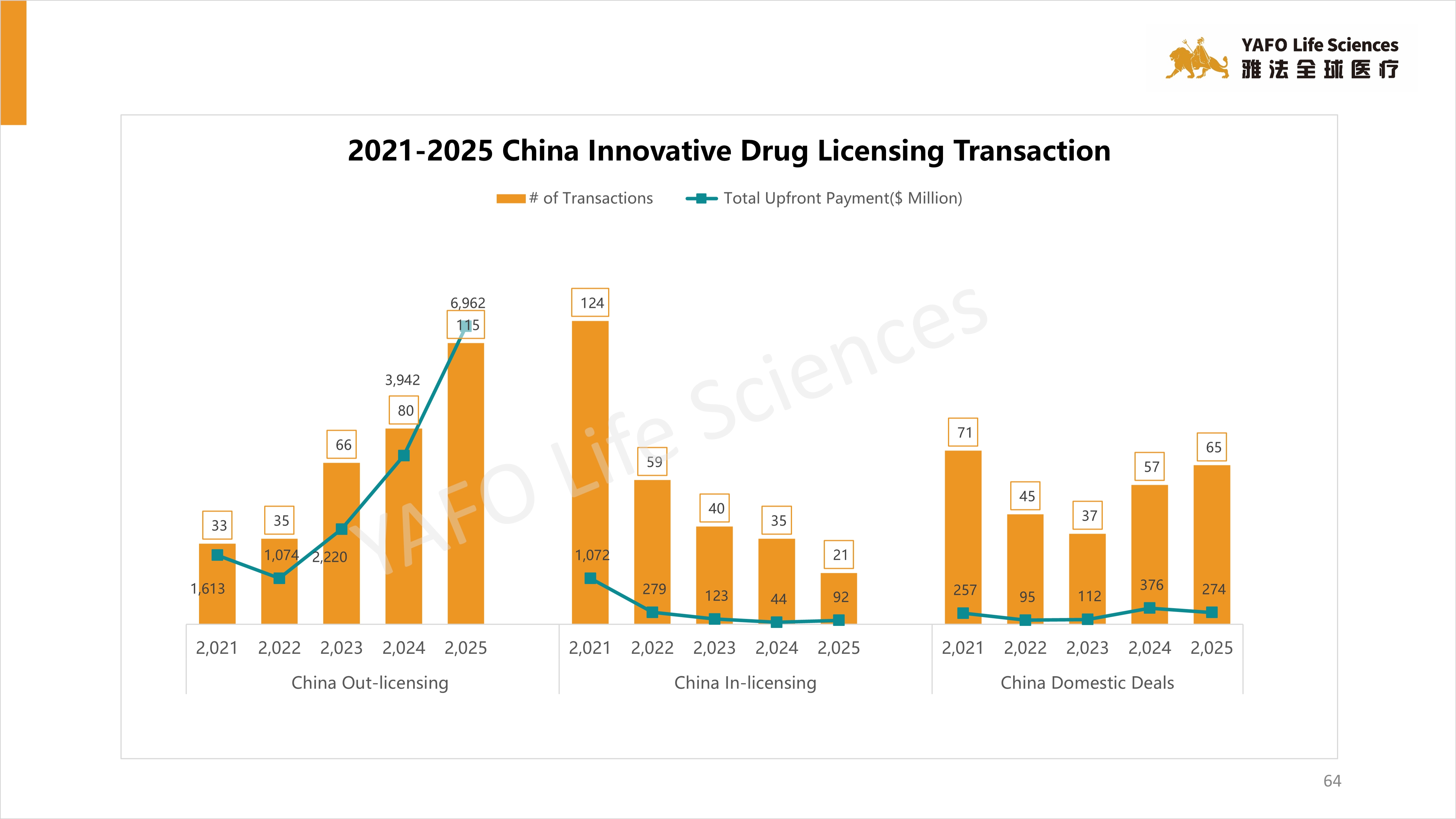
4. 2019-2024 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。