1. Summary of the Week
From April 11 to 17, a total of 21 agreements were signed worldwide. Within China’s biotech industry, there were four out-licensing deals, one in-licensing deal, and five domestic transactions.
The top in-licensing deal in China was between Aligos Therapeutics and Amoytop Biotech for the Phase II asset pevifoscorvir, with an upfront payment of $25 million and a total deal value of $445 million. The leading out-licensing deal was between Haisco Pharmaceutical and AbbVie, covering the Phase II and preclinical assets HSK55718 and HSK51155, with an upfront payment of $30 million and a total deal value of $745 million.
Globally, 11 deals were announced. The largest transaction was between Telix Pharmaceuticals and Regeneron Pharmaceuticals for preclinical diagnostic assets and radiopharmaceutical therapy programs, with an upfront payment of $40 million.
2026年4月11日-17日,全球医药市场共签署了21项资产授权和合作协议。中国市场共达成10项交易,包括4项出海交易、1项引进交易和5项国内交易。
国内市场上,本周最大的引进交易是特宝生物从Aligos Therapeutics引进临床二期资产pevifoscorvir,首付款2500万美元,总金额4.45亿美元。最大的出海交易是海思科与Abbvie就临床二期资产HSK55718和临床前资产HSK51155达成授权合作,首付款3000万美元,总金额7.45亿美元。
国际市场上,本周共签署了11项资产授权和合作协议。最大的交易是TelixPharmaceuticals与Regeneron Pharmaceuticals就临床前阶段放射性诊断和治疗药物达成合作,首付款4000万美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China IL |
Aligos Therapeutics | Amoytop Biotech 特宝生物 |
pevifoscorvir | chronic hepatitis B | Phase II | 445 | 25 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China OL |
Haisco Pharmaceutical 海思科 |
AbbVie | HSK55718; HSK51155 | post-operative pain; acute pain | Phase II; Pre-Clinical | 745 | 30 | United States; Europe; Japan; Other |
| China OL |
Evive Biotech 亿一生物 |
Molteni Farmaceutici | efbemalenograstim alfa | chemotherapy-induced neutropenia; febrile neutropenia; Parkinson’s disease | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Qilu Pharmaceutical 齐鲁制药 |
Mochida Pharmaceutical | follitropin alfa biosimilar | female infertility; ART; FSH deficiency; embryo transfer; PCOS; anovulation; IVF; LH deficiency | Approved | Undisclosed | Undisclosed | Japan |
| China OL |
Hybio Pharmaceutical 翰宇药业 |
MCG Pharma | semaglutide biosimilar; liraglutide biosimilar | overweight; type 2 diabetes; cardiovascular disease; obesity | Phase III; Approved | Undisclosed | Undisclosed | Other |
| China Domestic |
Bio-Thera Solutions 百奥泰 |
Lepu Pharmaceuticals 乐普药业 |
bevifibatide | unstable angina; ACS; myocardial infarction; ischemic stroke; thrombotic diseases | Approved | 63 | Undisclosed | Global |
| China Domestic |
Capital Medical University 首都医科大学 |
Gloria Pharmaceuticals 誉衡药业 |
异喹啉类衍生物 | neurodegenerative disease | Pre-Clinical | 2 | Undisclosed | Global |
| China Domestic |
Insilico Medicine 英矽智能 |
Ancestorcell 昂昇生物 |
stem cell–derived exosome therapy | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
Institute of Medicinal Biotechnology, CAMS 中国医学科学院医药生物技术研究所 |
SunNovo 阳光诺和 |
IMB0304 | bacterial infection | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
AilsynBio 昂心生物 |
Dong-E-E-Jiao 东阿阿胶 |
POI program; AI platform | premature ovarian insufficiency | Undisclosed | Undisclosed | Undisclosed | Global |
2a. China Section
2b. Global Section
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation; Option | Telix Pharmaceuticals | Regeneron Pharmaceuticals | diagnostic assets; radiopharmaceutical therapy programs | oncology | Pre-Clinical | Undisclosed | 40 | Global |
| License | QIMR Berghofer Medical Research Institute | Kazia Therapeutics | SETDB1 platform; MSETC | oncology | Pre-Clinical | 1.4 | Undisclosed | Global |
| License | Mayo Clinic | Mesoblast | CAR-MSC platform | oncology | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Quantori | ImmuVia | Q-Scientist platform; IMV-M | NSCLC; pancreatic cancer; cancer; gynecological cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | BostonGene | ImmunoGenesis | IMGS-001; AI multi-omics platform | TNBC; esophageal cancer; OVC; CRC; solid tumor; prostate cancer; gastric cancer; bladder cancer | Phase I | Undisclosed | Undisclosed | Global |
| Cooperation | Ramot at Tel Aviv University | Galmed Pharmaceuticals | Aramchol | PSC; MAFLD; MASH; lipodystrophy; gastrointestinal cancer | Phase III | Undisclosed | Undisclosed | Global |
| Cooperation | Leo Pharma | Er-Kim | toripalimab | Infection; Hematology; Rare Disease; Dermatology; Oncology; Otolaryngology | Approved | Undisclosed | Undisclosed | Other |
| Cooperation; License | Eli Lilly | Vasa Therapeutics | TuneLab platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Genvor | Canlab International | natural peptide candidates | overweight; hair loss | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; Investment; Option | Sana Biotechnology | Mayo Clinic | SC451 | type 1 diabetes | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Ewha Womans University | Sanjin Pharma | CNS treatment | neurological disorder; migraine | Pre-Clinical | Undisclosed | Undisclosed | Global |
3. M&A Deals
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) | Upfront ($ million) |
|---|---|---|---|---|---|---|
| Proposed | CrossBridge Bio | Eli Lilly | Full Acquisition | CB-120 | 300 | Undisclosed |
4. Top Deals of 2026
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| 2026-01-30 | CSPC Pharma 石药集团 |
AstraZeneca | Endocrine & Metabolic | IND; Pre-Clinical | 18,500 | 1,200 |
| 2026-01-13 | RemeGen 荣昌生物 |
AbbVie | Rare Disease; Oncology | Phase II | 5,600 | 650 |
| 2026-02-08 | Innovent Biologics 信达生物 |
Eli Lilly | Immunology; Oncology | Pre-Clinical | 8,850 | 350 |
| 2026-02-02 | Sanegene Bio 圣因生物 |
Genentech Roche |
Unknown/To be determined | Pre-Clinical | 1,700 | 200 |
| 2026-01-12 | SciNeuro Pharma 赛神医药 |
Novartis | Neurology | Pre-Clinical | 1,665 | 165 |
| 2026-03-04 | CTTQ Pharma 正大天晴 |
Sanofi | Hematology; Rare Disease; Immunology; Oncology | Approved | 1,530 | 135 |
| 2026-03-29 | Insilico Medicine 英矽智能 |
Eli Lilly | Endocrine & Metabolic; Unknown/To be determined | Pre-Clinical | 2,750 | 115 |
| 2026-01-09 | Hiasco 海思科 |
AirNexis Therapeutics | Respiratory; Infection | Phase II | 1,063 | 108 |
| 2026-02-13 | GenHouse 勤浩医药 |
Gilead Sciences | oncology | Phase I | 1,530 | 80 |
| 2026-02-05 | Henlius Biotech 复宏汉霖 |
Eisai | Gastroenterology; Infection; Rare Disease; Hematology; Immunology; Oncology; Endocrine & Metabolic | Approved | 388.31 | 75 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| 2026-04-17 | Aligos Therapeutics | Amoytop Biotech 特宝生物 |
Infection; Gastroenterology | Phase II | 445 | 25 |
| 2026-03-09 | Rapport Therapeutics | Tenacia Biotechnology 元羿生物 |
Neurology; Psychiatry | Phase II | 328 | 20 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| 2026-03-23 | Corxel Pharmaceuticals 箕星药业 |
Everest Medicines 云顶新耀 |
Cardiovascular | Approved | 50 | 30 |
| 2026-02-05 | Micot Technology 麦科奥特 |
Everest Medicines 云顶新耀 |
Genitourinary; Endocrine & Metabolic | Phase III | 172.48 | 27.82 |
| 2026-01-17 | Raynovent 众生药业 |
Qilu Pharmaceutical 齐鲁制药 |
Gastroenterology; Respiratory; Endocrine & Metabolic | Phase III | 139.10 | 27.82 |
| 2026-01-12 | Wan Bang De 万邦德 |
Hisoar Pharmaceutical 海翔药业 |
Neurology; Rare Disease | Pre-Clinical | 20.87 | 16.69 |
| 2026-01-06 | Accendatech 尚德药缘 |
Yifan Pharmaceutical 亿帆医药 |
Neurology; Ophthalmology; Respiratory; Hematology; Rare Disease; Immunology; Oncology | Phase III | 20.87 | 13.91 |
| 2026-02-04 | Akeso Biopharma; Kangrong Dongfang 康方生物 |
Jumpcan Pharmaceutical 济川药业 |
Rare Disease; Cardiovascular | Approved | 12.52 | 11.13 |
| 2026-01-20 | Insilico Medicine 英矽智能 |
Hengtai Biotechnology 衡泰生物 |
Neurology | Pre-Clinical | 66 | 10 |
| 2026-01-30 | PrimeGene 普祺医药 |
Jumpcan Pharmaceutical 济川药业 |
Rare Disease; Immunology; Dermatology; Otolaryngology | Phase III | 13.91 | 5.56 |
| Date | Licensor | Licensee | Field | Stage | Total amount ($ million) |
Upfront ($ million) |
|---|---|---|---|---|---|---|
| 2026-03-20 | Synnovation Therapeutics | Novartis | Oncology | Phase I/II | 3,000 | 2,000 |
| 2026-03-19 | Corium Therapeutics | Collegium Pharmaceutical | Psychiatry | Approved | 785 | 650 |
| 2026-03-09 | GSK | Alfasigma | Gastroenterology; Rare Disease; Dermatology; Endocrine & Metabolic | Filed | 690 | 300 |
| 2026-02-23 | Vir Biotechnology | Astellas Pharma | Oncology | Phase I/II | 1,705 | 240 |
| 2026-03-23 | Kali Therapeutics | Sanofi | Musculoskeletal; Immunology | Phase I | 1,230 | 180 |
| 2026-02-18 | CSL | Eli Lilly | Respiratory; Gastroenterology; Infection; Rare Disease; Musculoskeletal; Immunology; Cardiovascular; Oncology; Genitourinary; Endocrine & Metabolic | Phase III | Undisclosed | 100 |
| 2026-02-18 | Unnatural Products | Novartis | Cardiovascular | Pre-Clinical | 1,800 | 100 |
| 2026-01-29 | Repertoire Immune Medicines | Eli Lilly | Immunology | Pre-Clinical | 1,925 | 85 |
| 2026-04-09 | Kymera Therapeutics | Gilead Sciences | Oncology | Pre-Clinical | 750 | 85 |
| 2026-01-12 | Nuvation Bio | Eisai | Rare Disease; Oncology | Approved | 230 | 60 |
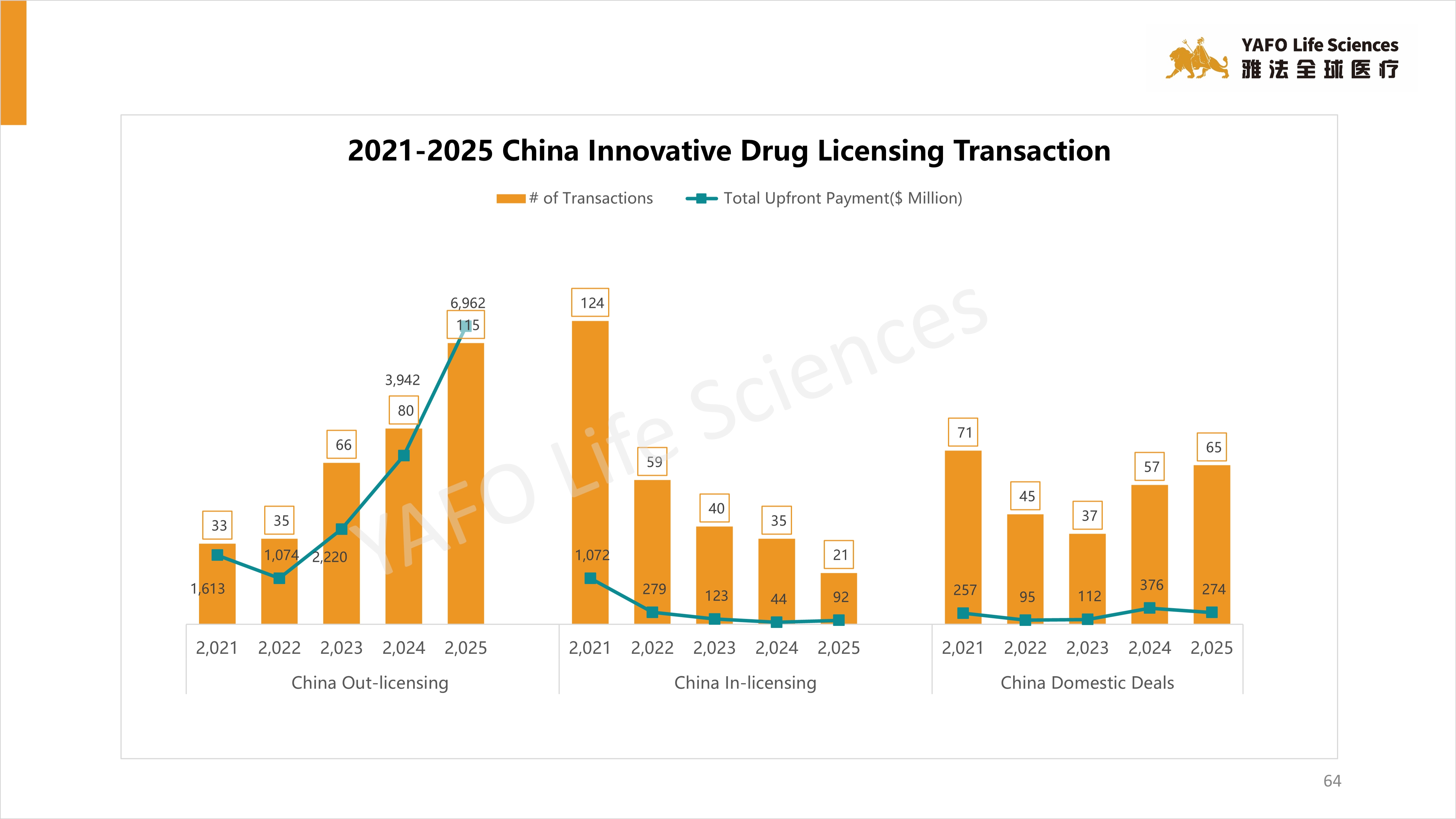
5. 2021-2025 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。