1. Executive Summary of the Month
In April 2026, 103 biopharmaceutical agreements were announced worldwide. In China’s biotech sector, there were 17 out-licensing deals, 5 in-licensing deals, and 11 domestic partnerships.
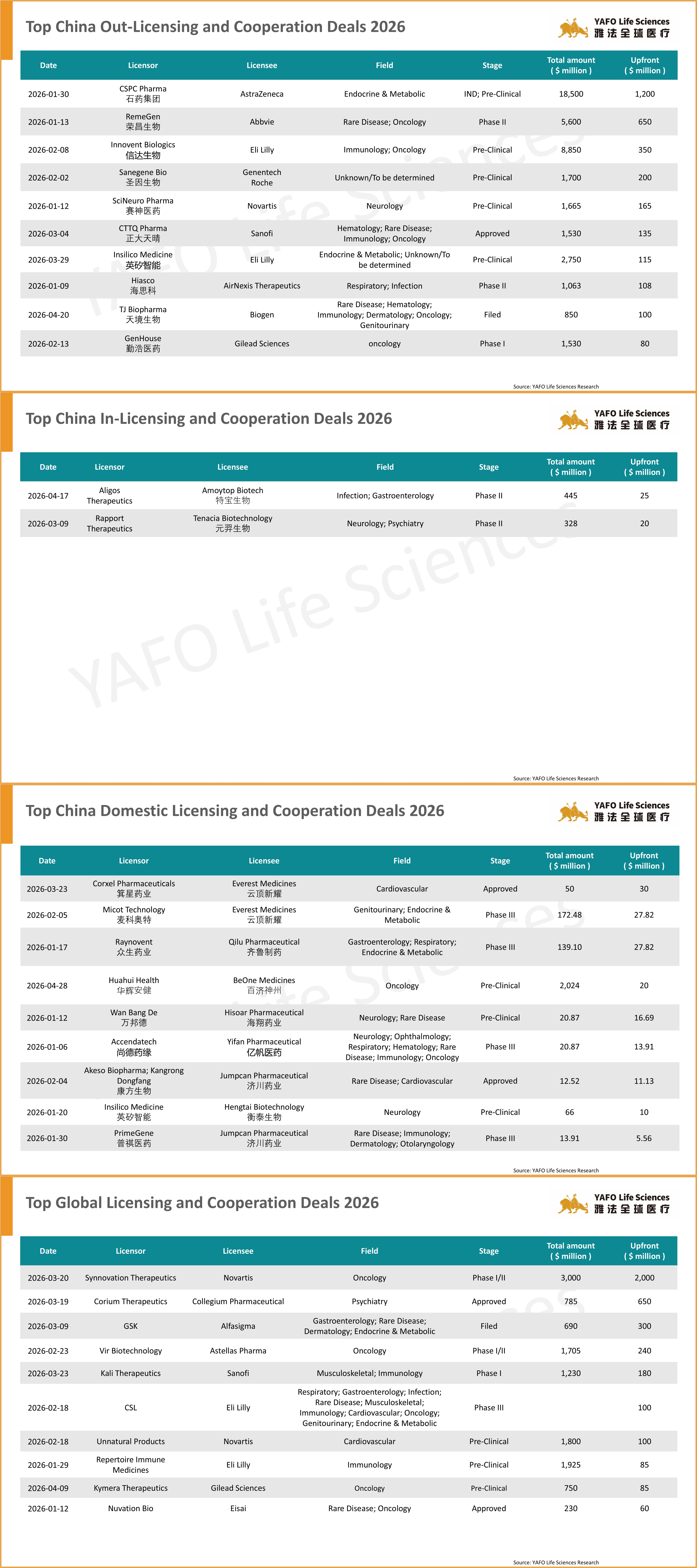
In China, the largest in-licensing deal involved Aligos Therapeutics and Amoytop Biotech for the Phase II asset pevifoscorvir, with an upfront payment of $25 million and a total deal value of $445 million. The largest out-licensing deal was signed by TJ Biopharma and Biogen, with an upfront payment of $100 million and a total deal value of $850 million, for the filed asset felzartamab. The largest domestic deal was between Huahui Health and BeOne Medicines for the pre-clinical asset HH160, including an upfront payment of $20 million and a total deal value of $2.02 billion.
Internationally, 70 cross-border deals were reported, with the largest being the agreement between Kymera Therapeutics and Gilead Sciences for a pre-clinical asset KT-200, with an upfront payment of $85 million and a total deal value of $750 million.
2026年4月,全球医药市场共签署了103项资产授权和合作协议。中国市场共达成33项交易,包括17项出海交易、5项引进交易和11项国内交易。
国内市场上,本月最大的引进交易是特宝生物从Aligos Therapeutics引进临床二期资产pevifoscorvir,首付款2500万美元,总金额4.45亿美元;最大的出海交易是天境生物和Biogen就已获批资产felzartamab达成授权合作,首付款1亿美元,总金额可达8.5亿美元;最大的国内交易是百济神州获得华辉安健临床前资产HH160的独家选择权,首付款2000万美元,总金额可达20.2亿美元。
国际市场上,本月共签署了70项资产授权和合作协议。最大的交易是Gilead Sciences行使选择权,从Kymera Therapeutics引进临床前资产KT-200,首付款8500万美元,总金额可达7.5亿美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China IL |
Aligos Therapeutics | Amoytop Biotech 特宝生物 |
pevifoscorvir | chronic hepatitis B | Phase II | 445 | 25 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China IL |
Pharmacosmos | China Medical System 康哲药业 |
ferric derisomaltose; iron dextran | chronic renal anemia; chemotherapy-induced anemia; postpartum hemorrhage; gastrointestinal disease; hypermenorrhea; IDA; anemia; postpartum anemia; RLS | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL |
Johnson & Johnson | Keyuan Trade 科园贸易 上海医药 |
Invega Hafyera paliperidone palmitate; Invega Trinza | bipolar I disorder; dementia-related psychosis; schizophrenia | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL |
AbbVie | Santen Pharmaceutical (China) 参天中国 |
bimatoprost; bimatoprost+timolol; brimonidine+timolol; brimonidine | glaucoma or ocular hypertension; eyelash hypotrichosis; alopecia areata; diabetic retinopathy; AMD; macular edema; DME; geographic atrophy; rosacea; NAION | Approved | Undisclosed | Undisclosed | Chinese mainland |
| China IL |
Minophagen Pharmaceutical | Main Luck Pharmaceuticals 万乐药业 |
bexarotene | Neurology; Hematology; Rare Disease; Immunology; Dermatology; Psychiatry; Oncology; Endocrine & Metabolic | Approved | Undisclosed | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan |
| China OL |
TJ Biopharma 天境生物 |
Biogen | felzartamab | idiopathic membranous nephropathy (IMN); IgA nephropathy (IgAN); renal transplant rejection; antibody-mediated rejection; lupus nephritis (LN); membranous nephropathy; multiple myeloma (MM); systemic lupus erythematosus (SLE) | Filed | 850 | 100 | Chinese mainland; Hong Kong, Macao and Taiwan |
| China OL |
Haisco Pharmaceutical 海思科 |
AbbVie | HSK55718; HSK51155 | post-operative pain; acute pain | Phase II; Pre-Clinical | 745 | 30 | United States; Europe; Japan; Other |
| China OL |
United BioPharma; Ascendant Health 联合生物 |
Cue Biopharma | UB-221 | autoimmune disease; chronic spontaneous urticaria (CSU) | Phase II | 692 | 15 | United States; Europe; Japan; Other |
| China OL |
Gan Lee Pharmaceuticals 甘李药业 |
JW Pharmaceutical | bofanglutide | obesity; type 2 diabetes; overweight; obstructive sleep apnea (OSA) | Phase III | 81 | 5 | Other |
| China OL |
Xcovery 贝达药业 |
EVERSANA | ensartinib | Rare Disease; Hematology; Oncology | Approved | Undisclosed | Undisclosed | United States |
| China OL |
Mabwell Bioscience 迈威生物 |
Undisclosed | denosumab biosimilar | breast cancer; osteoporosis; bone cancer; giant cell tumor of bone; multiple myeloma (MM); bone metastasis | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Formosa Pharmaceuticals 台新药 |
SERI | next-generation ophthalmic formulations | ophthalmology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China OL |
Fosun Pharmaceutical 复星医药 |
Arcera | Advanced technology cooperation; asset licensing; neuroscience strategic cooperation | neurodegenerative disease; Alzheimer’s disease | Undisclosed | Undisclosed | Undisclosed | Global |
| China OL |
Adcoris 爱科瑞思 |
K2 Therapeutic | ACR246; bispecific antibody ADC project | NSCLC; solid tumor; ovarian cancer (OVC); pancreatic cancer; breast cancer; CRC; cervical cancer; prostate cancer; kidney cancer; esophageal cancer | Phase I/II; Pre-Clinical | Undisclosed | Undisclosed | Global |
| China OL |
Xuanzhu Biopharm 轩竹生物 |
Boston Oncology | bireociclib; dirozalkib | brain cancer; breast cancer; hormone receptor positive breast cancer; prostate cancer; solid tumor; NSCLC; cancer; lung cancer; oncology | Approved | Undisclosed | Undisclosed | Other |
| China OL |
WuXi XDC 药明合联 |
Akari Therapeutics | PH1 payload | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| China OL |
Evive Biotech 亿一生物 |
INTSEL CHIMOS | efbemalenograstim alfa | chemotherapy-induced neutropenia (CIN); febrile neutropenia; neutropenia; Parkinson’s disease (PD) | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Evive Biotech 亿一生物 |
Molteni Farmaceutici | efbemalenograstim alfa | chemotherapy-induced neutropenia; chemotherapy induced febrile neutropenia; Parkinson’s disease | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Qilu Pharmaceutical 齐鲁制药 |
Mochida Pharmaceutical | follitropin alfa biosimilar | female infertility; ART; FSH deficiency; embryo transfer; PCOS; anovulation; IVF; LH deficiency | Approved | Undisclosed | Undisclosed | Japan |
| China OL |
Hybio Pharmaceutical 翰宇药业 |
MCG Pharma | semaglutide biosimilar; liraglutide biosimilar | overweight; type 2 diabetes; diabetic cardiovascular disease; obesity | Phase III; Approved | Undisclosed | Undisclosed | Other |
| China OL |
Hybio Pharmaceutical 翰宇药业 |
Torrent Pharmaceuticals | lanreotide acetate pre-filled sustained-release injection solution | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | United States |
| China OL |
Hansoh Pharmaceutical 翰森制药 |
Tortugas Neuroscience | HS-10380; HS-10353 | schizophrenia; Alzheimer’s disease agitation; bipolar I disorder; tinnitus; depressive disorder; PPD; MDD | Phase II | Undisclosed | Undisclosed | Global |
| China Domestic |
Huahui Health 华辉安健 |
BeOne Medicines 百济神州 |
HH160 | cancer | Pre-Clinical | 2,024 | 20 | Global |
| China Domestic |
Wenzhou Medical University 温州医科大学 |
Shapuaisi Pharmaceutical 莎普爱思 |
SPAS-26021 | myopia | Pre-Clinical | 10 | 1 | Global |
| China Domestic |
Bio-Thera Solutions 百奥泰 |
Lepu Pharmaceuticals 乐普药业 |
bevifibatide | unstable angina; ACS; myocardial infarction; ischemic stroke; thromboangiitis obliterans; NSTEMI; thrombotic diseases | Approved | 63 | Undisclosed | Global |
| China Domestic |
Hansoh Pharmaceutical 翰森制药 |
Hengrui Pharmaceuticals 恒瑞医药 |
KiOmedine vs One (CM-Chitosan) | osteoarthritis (OA) | Approved | 18 | Undisclosed | Chinese mainland; Hong Kong, Macao and Taiwan |
| China Domestic |
Capital Medical University 首都医科大学 |
Gloria Pharmaceuticals 誉衡药业 |
Isoquinoline derivatives | neurodegenerative disease | Pre-Clinical | 2 | Undisclosed | Global |
| China Domestic |
Boji Medical 博济医药 |
Pusch Pharmaceutical 普什制药 |
FCZR (fluconazole ear drops) | otomycosis | IND | 2 | Undisclosed | Global |
| China Domestic |
Jacobson Pharma 雅各臣科研制药 |
Weijian Pharmaceutical 维健医药 |
ARSENOL (arsenic trioxide oral solution) | acute promyelocytic leukemia (APL) | Phase II | Undisclosed | Undisclosed | Chinese mainland |
| China Domestic |
Biocytogen 百奥赛图 |
Sihuan Pharmaceutical 四环医药 |
innovative therapies | Overweight | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
Insilico Medicine 英矽智能 |
Ancestorcell 昂昇生物 |
stem cell–derived exosome therapy | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
The Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences 中国医学科学院医药生物技术研究所 |
SunNovo 阳光诺和 |
IMB0304 | bacterial infection | Pre-Clinical | Undisclosed | Undisclosed | Global |
| China Domestic |
AilsynBio 昂心生物 |
Dong-E-E-Jiao 东阿阿胶 |
POI program; AI drug discovery platform | premature ovarian insufficiency | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | Kymera Therapeutics | Gilead Sciences | KT-200 | breast cancer; solid tumor | Pre-Clinical | 750 | 85 | Global |
| Cooperation; Option | Telix Pharmaceuticals | Regeneron Pharmaceuticals | diagnostic assets; radiopharmaceutical therapy programs | oncology | Pre-Clinical | Undisclosed | 40 | Global |
| Asset Acquisition | Assertio Holdings | Cosette Pharmaceuticals | Sympazan; indomethacin; methotrexate; Zipsor; Cambia; ketorolac | Neurology; Rare Disease; Musculoskeletal; Immunology; Endocrine & Metabolic; Anesthesia & Analgesia; Cardiovascular; Ophthalmology; Hematology; Oncology; Dermatology | Approved | Undisclosed | 35 | Global |
| Asset Acquisition | Johnson & Johnson | MeiraGTx | botaretigene sparoparvovec | X-linked retinitis pigmentosa (XLRP) | Phase III | Undisclosed | 25 | Global |
| Cooperation; Option | C4 Therapeutics | Roche | TORPEDO DACs | oncology | Pre-Clinical | 1,020 | 20 | Global |
| License | Scohia Pharma | Neurocrine Biosciences | SCO-483 | congenital adrenal hyperplasia (CAH); Cushing’s syndrome | Pre-Clinical | 570 | 20 | Global |
| License | Dyno Therapeutics | Astellas Pharma | CapsidMap™ platform; gene therapy | musculoskeletal disorder; cardiomyopathy | Pre-Clinical | Undisclosed | 15 | Global |
| License | Halozyme Therapeutics | Vertex Pharmaceuticals | Hypercon™ technology | Undisclosed | Undisclosed | Undisclosed | 15 | Global |
| License; Cooperation | NEC | Transgene | TG4050; AI-based neoantigen prediction platform | peritoneal cancer; SCCHN; fallopian tube cancer; head and neck cancer; ovarian cancer (OVC) | Phase I/II | Undisclosed | 6 | Global |
| Cooperation; License | Profluent | Eli Lilly | AI-designed recombinases; recombinase-based gene editing therapeutics | unknown/to be determined | Pre-Clinical | 225 | Undisclosed | Global |
| License | PineTree Therapeutics | AstraZeneca | PTX-299 | cancer | Pre-Clinical | 500 | Undisclosed | Global |
| Divestiture | Dogwood Therapeutics | PRIDCor Therapeutics | famciclovir + celecoxib; valacyclovir + celecoxib | fibromyalgia; chronic fatigue syndrome (CFS); irritable bowel syndrome (IBS); COVID-19 | Phase II | 100 | Undisclosed | Global |
| License | QIMR Berghofer Medical Research Institute | Kazia Therapeutics | SETDB1-targeted epigenetic drug development platform; MSETC | oncology | Pre-Clinical | 1.4 | Undisclosed | Global |
| Cooperation | D&D Pharmatech | Pfizer | Formulation development of oral peptide dual agonist | obesity | Pre-Clinical | 1.3 | Undisclosed | Global |
| Cooperation | Sam Chun Dang | Undisclosed | semaglutide biosimilar | type 2 diabetes | Phase I | Undisclosed | Undisclosed | United States |
| Cooperation | BIOnSYSTEMS | ARKgen BioScions; LyseNTech | protein characterization and design platform; immune cell engager program | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global; Other |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| Cooperation | Candel Therapeutics | EVERSANA | aglatimagene besadenovec | pancreatic cancer; ovarian cancer (OVC); lung cancer; prostate cancer; mesothelioma; glioma; GBM; NSCLC; breast cancer | Phase III | Undisclosed | Undisclosed | United States |
| Cooperation | Undisclosed | Merck KGaA | Hybrid Drugs | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | United States |
| Cooperation | Miltenyi Biotec | CHA biotech | CHACAR-NK-201 | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | KM Biologics | SIIPL | KD-287 | Japanese encephalitis | Approved | Undisclosed | Undisclosed | Other |
| License | PharmAla Biotech | Undisclosed | APA-01 | traumatic brain injury (TBI); ischemic stroke | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | S.Biomedics | Parexel | TED-A9 | Parkinson’s disease (PD) | Phase I/II | Undisclosed | Undisclosed | United States; Japan |
| Cooperation | Atomatrix | inno.N | CANDDIE small molecules | obesity | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Noxopharm | Inhatarget Therapeutics | Sofra™ platform | pneumonitis | Undisclosed | Undisclosed | Undisclosed | Global |
| License | Apertura Gene Therapy | AviadoBio | TfR1 CapX™; AVB-406 | Alzheimer’s disease (AD) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Kestrel Therapeutics | AbbVie | KST-6051 | solid tumor; sarcoma; colorectal cancer (CRC); NSCLC; pancreatic ductal carcinoma | Phase I | Undisclosed | Undisclosed | Global |
| Asset Acquisition; Divestiture | Cumberland Pharmaceuticals | Apotex | lactulose; telavancin; conivaptan; Caldolor; Sancuso; Acetadote | Gastroenterology; Infection; Respiratory; Dermatology; Neurology; Rare Disease; Cardiovascular; Oncology; Toxicology | Approved | Undisclosed | Undisclosed | United States |
| License | MIT | Cyclo Therapeutics | adrabetadex | ischemic stroke; Niemann-Pick disease type C; pressure ulcers; AD; MGD | Filed | Undisclosed | Undisclosed | United States |
| Cooperation | Alteogen | GC Wellbeing | ALT-B4 | increasing tissue permeability | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | Touchlight | SVF Vaccines | doggybone DNA™ (dbDNA™) platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Tempus AI | Gilead Sciences | AI platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Alloy Therapeutics | Biogen | AntiClastic™ ASO Platform; ASO program | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Evofem Biosciences | Clovis Davis Pharmaceuticals | secnidazole | bacterial vaginosis; trichomoniasis; amebiasis; giardiasis; cholera; urethritis; E. coli infection | Approved | Undisclosed | Undisclosed | Other |
| Cooperation | FRONTEO | Astellas Pharma | drug discovery | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Yuhan | SK Chemicals | telmisartan + amlodipine + chlorthalidone | hypertension | Approved | Undisclosed | Undisclosed | Other |
| License | Istituto Gentili | Galena | medication used in paediatric care | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Other |
| Cooperation; Option | Interna Therapeutics | Daiichi Sankyo | Molecular Nano Motor (MNM) intracellular delivery platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | Keio University | DWTI | WP-1303 | glaucoma or ocular hypertension | Phase III | Undisclosed | Undisclosed | Global |
| Cooperation | Cincinnati Children’s Hospital Medical Center | BioOra | WZTL-002 | mantle cell lymphoma (MCL); DLBCL; B-cell acute lymphoblastic leukemia; PMBCL; follicular lymphoma (FL); B-cell lymphoma | Phase II | Undisclosed | Undisclosed | United States; Other |
| Cooperation; License | xFOREST Therapeutics | Nippon Shinyaku | FOREST technologies | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Rznomics | Surgenex | LNP-based RNA gene therapy | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Cartography Biosciences | Gilead Sciences | novel oncology target; ATLAS and SUMMIT Platforms | triple negative breast cancer (TNBC); non-small cell lung cancer (NSCLC) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; License | Scohia Pharma | Celltrion | CT-G32 | obesity | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Xeureka | Sumitomo Pharma | Small molecule drug candidates | Undisclosed | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | BioCubaFarma | Promomed | Multivalent therapeutic cancer vaccines | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Salipro Biotech | Undisclosed | Salipro® platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Takeda Pharmaceuticals | Axsome Therapeutics | balipodect | schizophrenia | Phase II | Undisclosed | Undisclosed | Global |
| License | Frontier Medicines | LG Chem | FMC-220 | cancer | Pre-Clinical | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| License | Mayo Clinic | Mesoblast | CAR-MSC technology platform | oncology | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Quantori | ImmuVia | Q-Scientist platform; IMV-M | non-small cell lung cancer (NSCLC); pancreatic cancer; cancer; gynecological cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | BostonGene | ImmunoGenesis | IMGS-001; AI-driven multi-omics platform | TNBC; esophageal cancer; ovarian cancer (OVC); colorectal cancer (CRC); solid tumor; prostate cancer; gastric cancer; bladder cancer | Phase I | Undisclosed | Undisclosed | Global |
| Cooperation | Acuitas Therapeutics | Circio | in vivo CAR-T cell therapy | unknown/to be determined | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Ramot at Tel Aviv University | Galmed Pharmaceuticals | Aramchol | PSC; gastrointestinal cancer; MAFLD; cholelithiasis; MASH; lipodystrophy | Phase III | Undisclosed | Undisclosed | Global |
| Cooperation | Leo Pharma | Er-Kim | toripalimab | Infection; Hematology; Rare Disease; Dermatology; Oncology; Otolaryngology | Approved | Undisclosed | Undisclosed | Other |
| Cooperation; License | Eli Lilly | Vasa Therapeutics | TuneLab platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Genvor | Canlab International | natural peptide candidates | Overweight; hair loss | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation; Investment; Option | Sana Biotechnology | Mayo Clinic | SC451 | type 1 diabetes | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Ewha Womans University | Sanjin Pharma | CNS treatment | disorder of nervous system; migraine | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Université Laval | Glycovax | galectin-3 inhibitor | fibrosis; renal fibrosis; idiopathic pulmonary fibrosis; cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Oncoceutics | HCR | dordaviprone | Hematology; Rare Disease; Oncology | Approved | Undisclosed | Undisclosed | United States |
| Cooperation | Galux | GC Biopharma | autoimmune antibody program | autoimmune disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Eisai | Tortugas Neuroscience | E2730; irsenontrine | epilepsy; photosensitive epilepsy; amyloidosis; Lewy body dementia; PRES; Parkinson disease dementia | Phase II | Undisclosed | Undisclosed | Global |
| Divestiture | AnaptysBio | First Tracks Biotherapeutics | ANB101; ANB033; rosnilimab | systemic sclerosis; SLE; eosinophilic esophagitis; celiac disease; ulcerative colitis; alopecia areata; rheumatoid arthritis | Pre-Clinical; Phase I; Phase II | Undisclosed | Undisclosed | Global |
| Cooperation | Acuitas Therapeutics | Genflow Biosciences | LNP delivery technology | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Galux | OliX Pharmaceuticals | AI-based siRNA delivery platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Entos Pharmaceuticals | L-CMD | LMNA-related CMD program | congenital muscular dystrophy (CMD) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | SynSmart | Amporin Pharmaceuticals | degenerative diseases therapy | neurodegenerative disease | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | NEXTGEN Bioscience | inno.N | NXC680 | idiopathic pulmonary fibrosis (IPF) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Cooperation | Barcode Nanotech | Galmed Pharmaceuticals | Aramchol | PSC; gastrointestinal cancer; MAFLD; cholelithiasis; MASH; lipodystrophy | Phase III | Undisclosed | Undisclosed | Global |
| Cooperation | Oxford BioTherapeutics | Bristol-Myers Squibb | OGAP-Verify based TCE therapy | oncology | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) | Upfront ($ million) |
|---|---|---|---|---|---|---|
| Proposed | Kelonia Therapeutics | Eli Lilly | Full Acquisition | KLN-1010 | 7,000 | 3,250 |
| Proposed | Tubulis | Gilead Sciences | Full Acquisition | TUB-030; TUB-040 | 5,000 | 3,150 |
| Proposed | Emalex Biosciences | Teva Pharmaceutical | Full Acquisition | ecopipam | 900 | 700 |
| Proposed | Neurona Therapeutics | UCB | Full Acquisition | rezanecel | 1,150 | 650 |
| Proposed | Hasten Biopharmaceuticals | Everest Medicines 云顶新耀 |
Full Acquisition | Undisclosed | 250 | 150 |
| Proposed | Replay | Leo Pharma | Full Acquisition | Undisclosed | Undisclosed | 50 |
| Proposed | Organon & Co. | Sun Pharma | Full Acquisition | benvitimod | 1,175 | Undisclosed |
| Proposed | Soleno Therapeutics | Neurocrine Biosciences | Full Acquisition | DCCR (diazoxide choline controlled release tablets) | 2,900 | Undisclosed |
| Proposed | Ajax Therapeutics | Eli Lilly | Full Acquisition | AJ1-11095 | 2,300 | Undisclosed |
| Proposed | KalVista Pharmaceuticals | Chiesi Farmaceutici | Full Acquisition | sebetralstat | 1,900 | Undisclosed |
| Proposed | Kestrel Therapeutics | AbbVie | Option Acquisition | KST-6051 | 450 | Undisclosed |
| Proposed | CrossBridge Bio | Eli Lilly | Full Acquisition | CB-120 | 300 | Undisclosed |
| Proposed | Assertio Holdings | Garda Therapeutics | Full Acquisition | eflapegrastim | 125 | Undisclosed |
| Proposed | Xinbai Pharmaceutical 新百药业 |
Double-crane Pharmaceutical 华润双鹤 |
Full Acquisition | Undisclosed | 33 | Undisclosed |
| Proposed | Galera Therapeutics | Obsidian Therapeutics | Merge | cytoTIL15 | Undisclosed | Undisclosed |
| Completed | Visufarma | Lupin | Full Acquisition | Undisclosed | Undisclosed | Undisclosed |
| Proposed | Cyclerion Therapeutics | Korsana Biosciences | Merge | KRSA-028 | Undisclosed | Undisclosed |
| Proposed | Kashiv BioSciences | Amneal Pharmaceuticals | Full Acquisition | Undisclosed | Undisclosed | Undisclosed |
2a. In-Licensing Deals
2b. Out-Licensing Deals
2c. Domestic Licensing Deals
3. Top Deals of the Year 2026
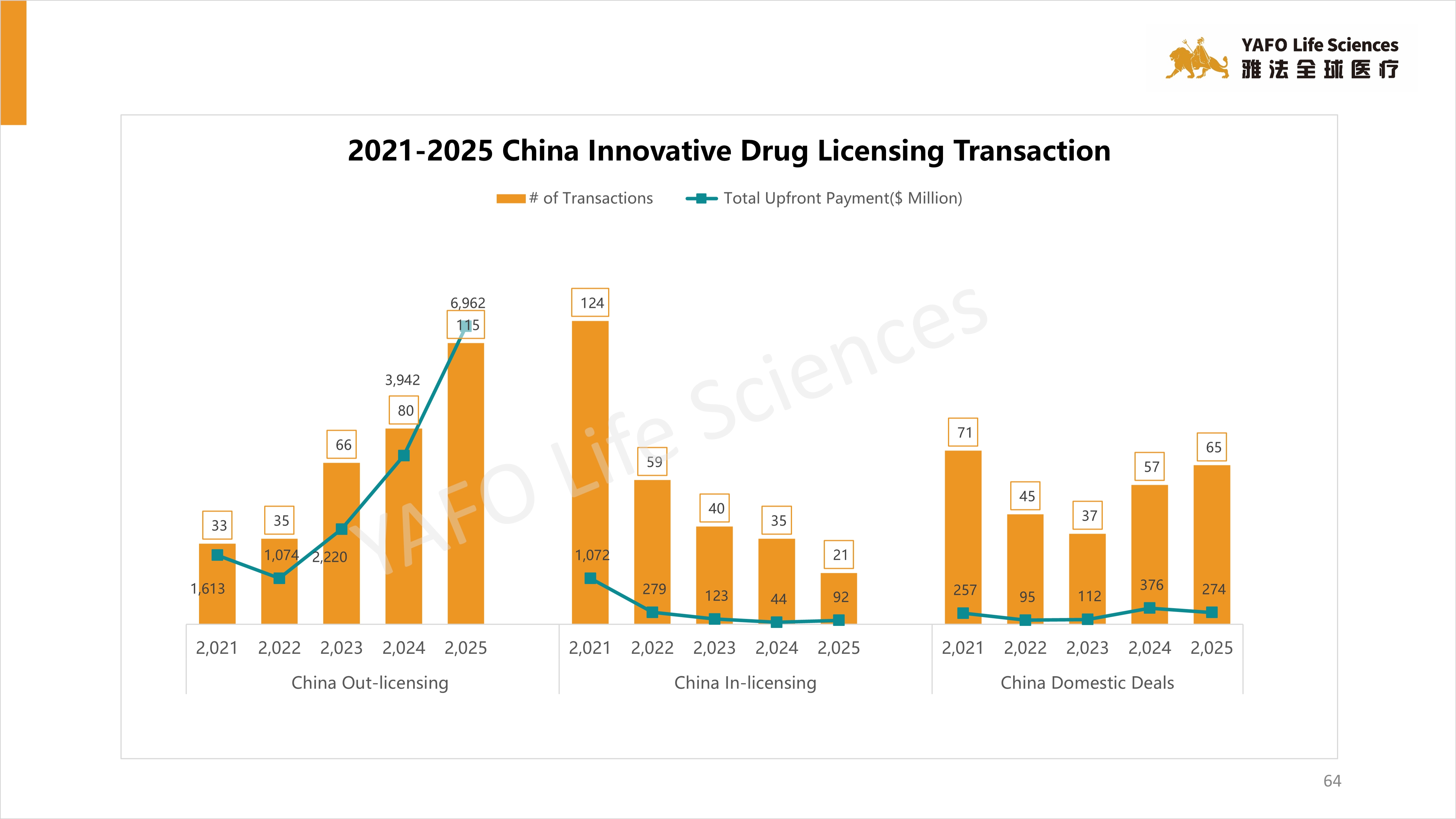
4. 2019-2024 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。