1. Summary of the Week
From May 1 to 8, a total of 20 agreements were signed worldwide. Within China’s biotech industry, there were two out-licensing deals, one in-licensing deal, and three domestic transactions.
The top licensing deal in China was between Siran Biotech and GSK, with a total deal value of $1 billion, for the Phase 1 asset SA030.
Globally, 14 deals were announced. The largest transaction was between BioCryst Pharmaceuticals and Neopharmed Gentili for Phase III asset navenibart, with an upfront payment of $70 million and a total deal value of 345 million.
2026年5月1日-8日,全球医药市场共签署了20项资产授权和合作协议。中国市场共达成6项交易,包括2项出海交易、1项引进交易和3项国内交易。
国内市场上,本周最大的出海交易是时安生物与GSK就临床一期siRNA资产SA030达成授权合作,总金额可达10亿美元。
国际市场上,本周共签署了14项资产授权和合作协议。最大的交易是BioCryst Pharmaceuticals与Neopharmed Gentili就临床三期资产navenibart达成授权合作,首付款7000万美元,总金额可达3.45亿美元。
2. Licensing Deals
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) |
Upfront ($ million) |
Deal Territory |
|---|---|---|---|---|---|---|---|---|
| China IL |
Kissei | Orient Europharma 友华生技 |
olutasidenib | Rare Disease; Hematology; Oncology | Approved | Undisclosed | Undisclosed | Other |
| China OL |
Siran Biotech 时安生物 |
GSK | SA030 | obesity; overweight | Phase I | 1,005 | Undisclosed | United States; Europe; Japan; Other |
| China OL |
Wellington Zhaotai; ZHAOTAI MEDICAL GROUP 昭泰医疗集团 |
BioOra | WZTL-002 | mantle cell lymphoma (MCL); diffuse large B-cell lymphoma (DLBCL); B-cell acute lymphoblastic leukemia; primary mediastinal B-cell lymphoma (PMBCL); follicular lymphoma (FL); B-cell lymphoma | Phase II | Undisclosed | Undisclosed | United States; Europe; Japan; Other |
| China Domestic |
InxMed 应世生物 |
Fosun Pharmaceutical 复星医药 |
ifebemtinib; IN10028 | Rare Disease; Oncology | Phase III; Phase I | Undisclosed | Undisclosed | Global; Chinese mainland |
| China Domestic |
HanchorBio 汉康生技 |
Hebei Tsinghua Development Institute 河北清华发展研究院 |
HCB101 | colorectal cancer (CRC); breast cancer; gastric cancer; non-Hodgkin lymphoma (NHL); triple negative breast cancer (TNBC); head and neck cancer; lung cancer; small cell lung cancer (SCLC); ovarian cancer (OVC); liver cancer; gastro-esophageal junction cancer; squamous cell carcinoma of the head and neck (SCCHN); solid tumor | Phase II | Undisclosed | Undisclosed | Global |
| China Domestic |
ChomiX Biotech 科络思生物 |
Sunshine Lake Pharma 东阳光药 |
Innovative drug research and development | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
2a. China Section
2b. Global Section
| Deal Type | Licensor | Licensee | Assets | Disease | Stage | Total amount ($ million) | Upfront ($ million) | Deal Territory |
|---|---|---|---|---|---|---|---|---|
| License | BioCryst Pharmaceuticals | Neopharmed Gentili | navenibart | hereditary angioedema (HAE) | Phase III | 345 | 70 | Europe |
| License | Arrowhead Pharmaceuticals | Madrigal Pharmaceuticals | JNJ-75220795 | metabolic dysfunction-associated steatohepatitis (MASH); fatty liver | Phase I | 1,000 | 25 | Global |
| Cooperation; License | Cancer Research UK; Cancer Research Horizons | UCB | novel cancer treatment | cancer | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License; Option | Therapeutics | Oragenics | PRV-002; cardiodialysis™ technology | traumatic brain injury (TBI) | Phase II | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Turn Biotechnologies | Daewoong Pharmaceutical | Intellectual property rights and assets related to the ERA technology platform | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| License; Cooperation | Eli Lilly | 1st Biotherapeutics | AI drug discovery platform | neurodegenerative disease; cancer | Undisclosed | Undisclosed | Undisclosed | Global |
| Option; License | Halozyme Therapeutics | GSK | ENHANZE® technology for oncology targets | oncology | Undisclosed | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Oncotelic | Lunai Bioworks | N2B (nose-to-brain) delivery system | Alzheimer’s disease (AD) | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Institute for Protein Innovation | Alloy Therapeutics | vhh (nanobody) libraries | Undisclosed | Undisclosed | Undisclosed | Undisclosed | Global |
| Cooperation | Apertura Gene Therapy | TSC Alliance | TfR1 CapX™; TSC gene therapy | tuberous sclerosis complex (TSC) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| Asset Acquisition | Matrivax | Elaris | MVX02 | Clostridium difficile infection (CDI) | Pre-Clinical | Undisclosed | Undisclosed | Global |
| License | Abeona Therapeutics | Sangrail Biologics | ABO-101 | mucopolysaccharidosis IIIB (MPSIIIB) | Phase I/II | Undisclosed | Undisclosed | Global |
| Cooperation | Kubota Vision | Laboratoires KÔL | emixustat | geographic atrophy; Stargardt disease; proliferative diabetic retinopathy | Phase III | Undisclosed | Undisclosed | Other |
| Cooperation; License | Halozyme Therapeutics | Oruka Therapeutics | ORKA-001; Hypercon™ technology | psoriasis; plaque psoriasis | Phase II | Undisclosed | Undisclosed | Global |
3. M&A Deals
| Status | Acquiree | Acquiror | Acquisition Type | Item | Total amount ($ million) | Upfront ($ million) |
|---|---|---|---|---|---|---|
| Proposed | Candid Therapeutics | UCB | Full Acquisition | GB261 | 2,200 | 2,000 |
| Proposed | Perfuse Therapeutics | Bayer | Full Acquisition | PER-001 | 2,450 | 300 |
| Proposed | Catalyst Pharmaceuticals | Angelini | Full Acquisition | vamorolone; perampanel; amifampridine | 4,100 | Undisclosed |
| Completed | DeuterOncology | Pathos | Partial Acquisition | OMO-2 | Undisclosed | Undisclosed |
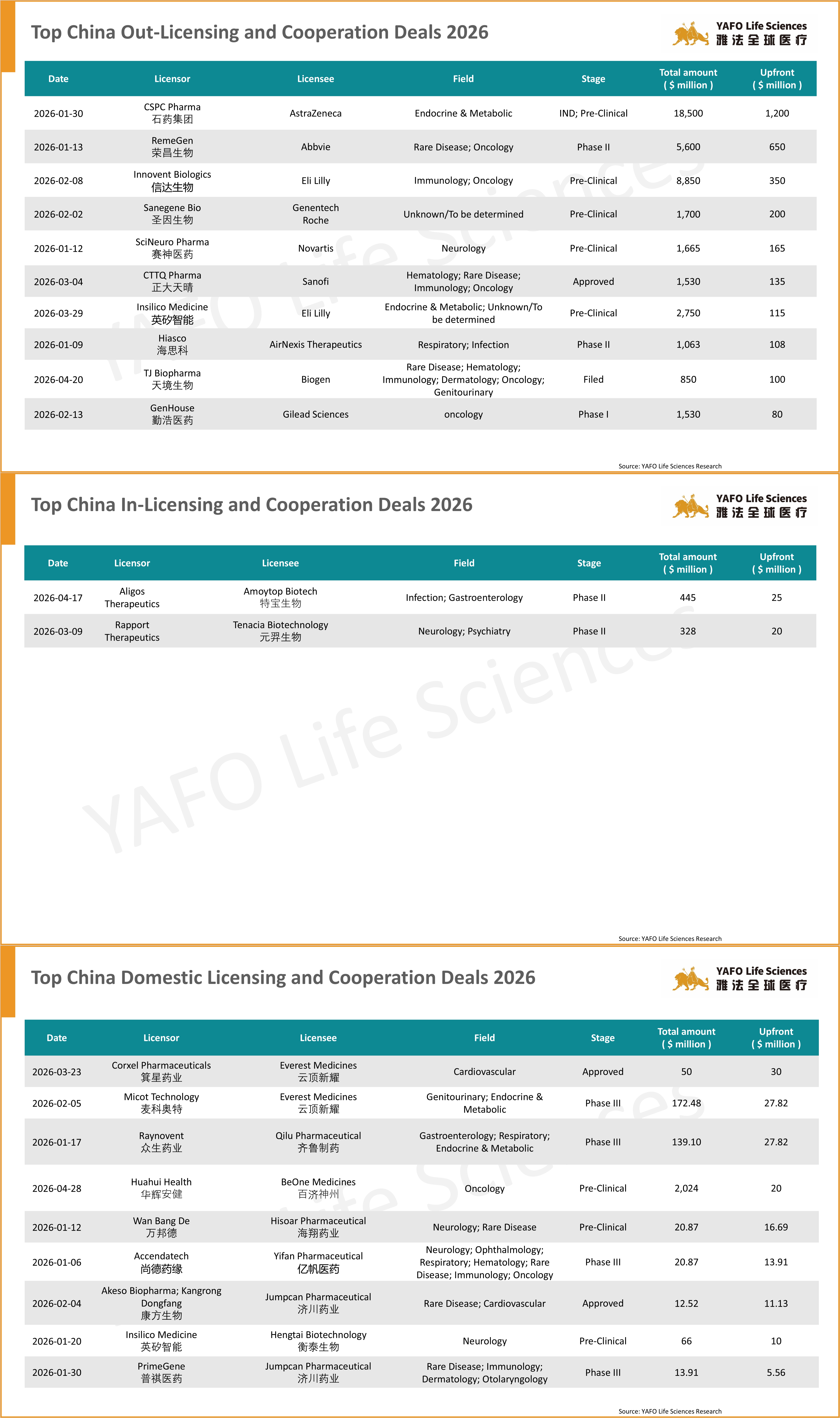
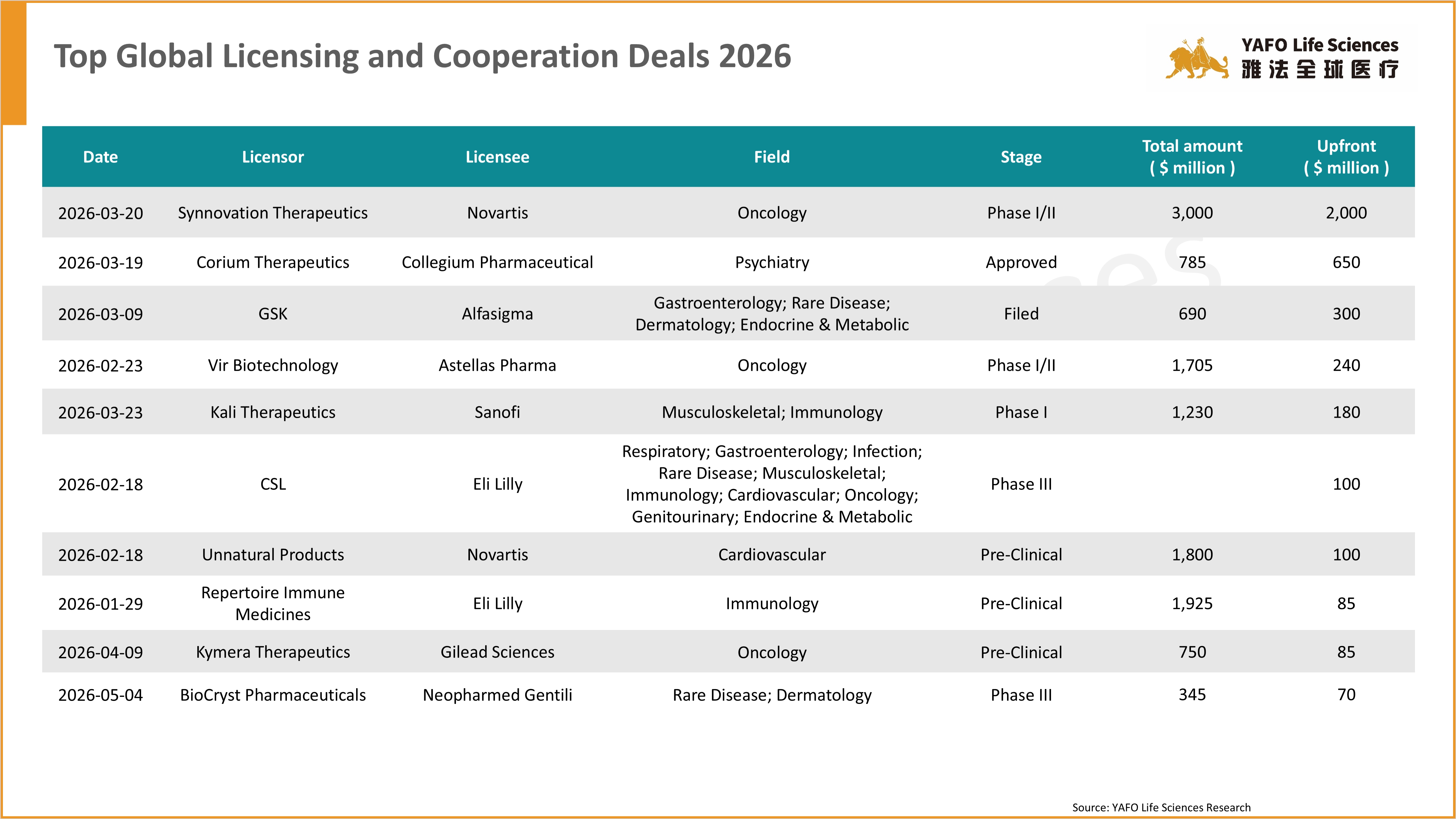
4. Top Deals of 2026
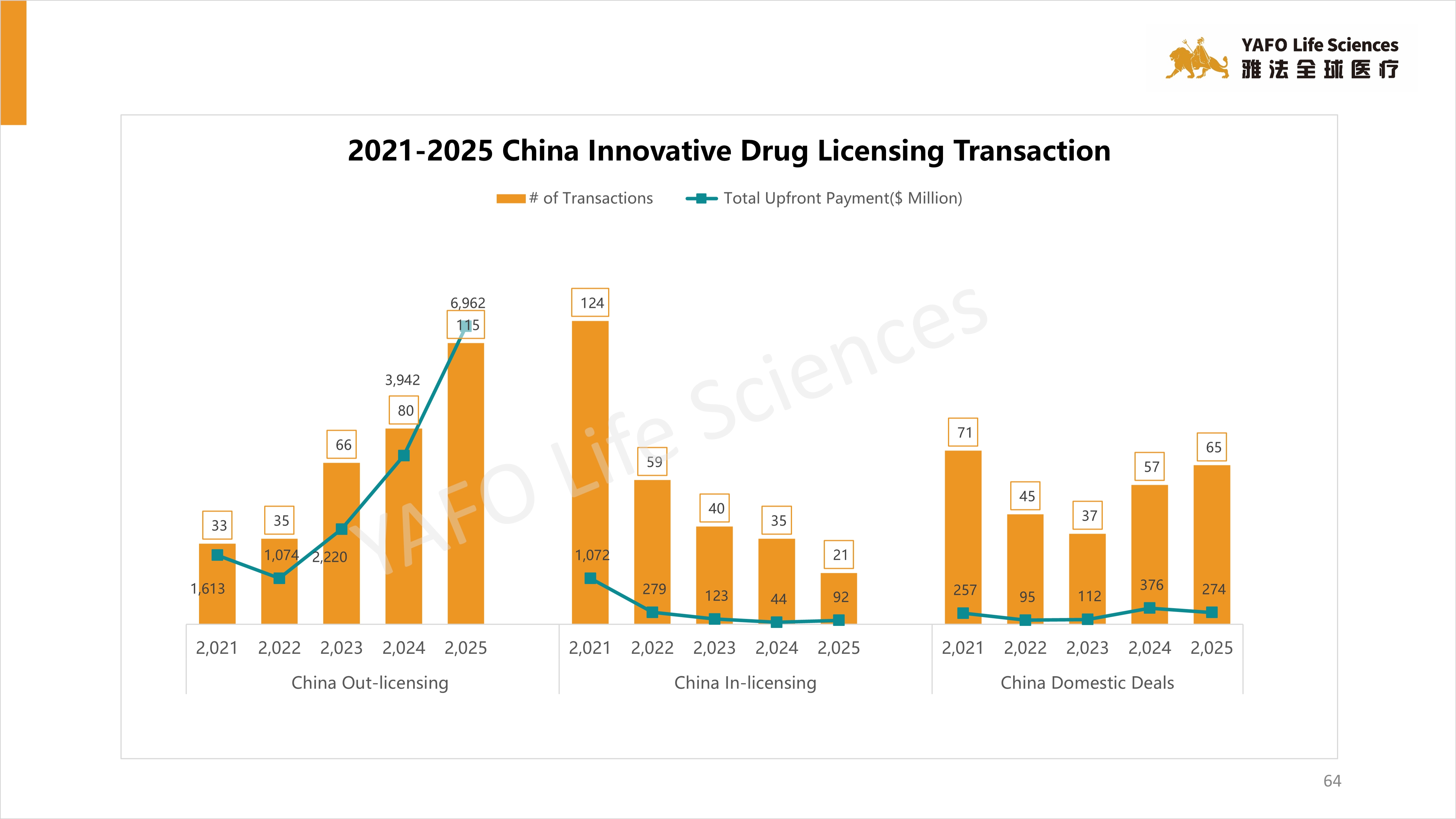
5. 2021-2025 China Innovative Drug Licensing Transactions
About YAFO Capital
Founded in 2013, YAFO Capital is a Shanghai-based boutique investment and advisory firm, with a professional team in our China, U.S., EU, and SEA offices. Partnering with Pharmaceutical companies, YAFO Fund mainly invests in global assets. YAFO Life Sciences is a leading advisory boutique focused on asset transactions. YAFO has built a strong, proven track record and closed dozens of in-licensing and out-licensing transactions with global pharma and biotech companies. Over the past five years, YAFO has been ranked as the No. 1 advisor for China cross-border licensing transactions. For more information, please visit http://www.yafocapital.com
雅法资本成立于2013年,作为新型投资和投行咨询机构,致力于中国及海外生物医药项目的投融资、资产跨境交易和资产孵化等。旗下雅法基金联合药企进行资产投资和并购,雅法全球医疗专注于医药产品跨境及国内授权交易。基于雅法在全球广泛的人脉与资源网络,在过去十年成功推进了大量的海外项目进入中国市场并协助多个中国产品完成海外授权。雅法拥有经验丰富的全球交易团队,覆盖美国、日本 、欧洲等全球主要医药创新区域。核心合伙人均为华尔街资深投行人士或具有跨国药企经历,为客户交易提供强力支持。雅法总部位于上海,在美国、欧洲、东南亚等地均设有分部。雅法在生物医药跨境授权及并购业务交易数量连续多年排名第一。